Quetiapine is a second-generation (atypical) antipsychotic. It’s approved by the FDA for schizophrenia, acute bipolar mania/mixed and depression, and bipolar maintenance (as an add-on to lithium or divalproex). The extended-release form is also approved as an add-on for depression.
Though its approved uses are limited, it’s widely used off-label as a single therapy for depression, anxiety, insomnia, OCD, and other conditions.
Quetiapine = Seroquel; Seroquel XR; Temprolide; Xeroquel; Ketipinor
PubChem: 5002
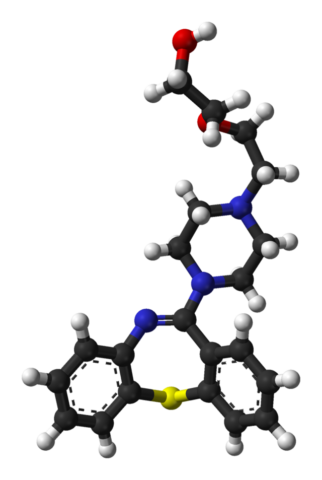
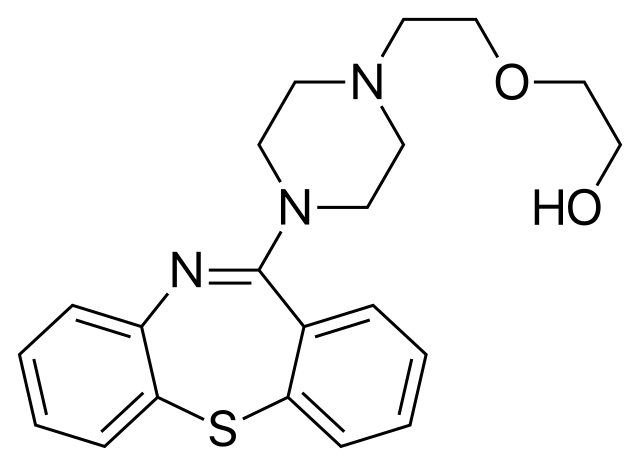
Molecular formula: C21H25N3O2S
Molecular weight: 383.51 g/mol
IUPAC: 2-[2-(4-benzo[b][1,4]benzothiazepin-6-ylpiperazin-1-yl)ethoxy]ethanol
Contents
Dose
Maximum recommended doses for adults:
- Schizophrenia: 750 mg/d
- Bipolar 1 (mania): 800 mg/d
- Bipolar depression: 300 mg/d
It’s usually given twice per day for psychotic disorders and bipolar disorder. Alternatively it may be given once daily if in extended-release form.
Timeline
Experience Reports
Effects
It’s primarily able to control psychotic symptoms, mood disorders, anxiety, and insomnia. Other uses have been explored, such as reducing substance abuse or reducing aggressiveness associated with a variety of conditions.
Common negatives (greater than 5% of patients and more than double the rate of placebo) include: dry mouth, dizziness, constipation, weakness, abdominal pain, sore throat, increased appetite, lethargy, and dyspepsia (indigestion).
Some patients report a general emotional blunting effect. It’s not known how common this is. For some people it’ll alleviate blunted emotion, such as in schizophrenia.
In the long-term there are concerns about significant weight gain, hyperglycemia (high glucose levels), hyperlipidemia (elevated lipids), and diabetes.
Like with many medications, it’s difficult to accurately describe its beneficial and adverse effects. Studies may be biased and high dropout rates lead to a lot of missing data. As such, a skeptical position is useful when evaluating the scientific literature.
The high dropout rate in studies and in clinical settings suggests noncompliance is an issue and that side effects or a lack of efficacy could be common.
Schizophrenia
Schizophrenia is a chronic psychiatric condition that produces positive symptoms (e.g. delusions, hallucinations), negative symptoms (e.g. low motivation, apathy, tiredness), and impaired cognition.
First-gen antipsychotics have been used for decades. They have proven efficacy for controlling positive symptoms, but they often have little to no effect on the negative and cognitive symptoms. In addition to a restricted efficacy profile, first-gen antipsychotics have a notable link to movement disorders like extrapyramidal symptoms (EPS) and tardive dyskinesia.
Second-gen antipsychotics are used with the intention of helping a larger set of schizophrenia symptoms while exposing people to a lower risk of movement disorders. There’s some evidence quetiapine and other drugs in this case are effective on those points.
Efficacy
Studies typically find quetiapine is significantly better than placebo and has an efficacy not too distinct from first-gen antipsychotics. Because its impact on core symptoms may not be reliably superior (example study: Buckley, 2004), individual differences and tolerability should guide prescribing.
There are some properties that could make it particularly useful in certain populations. Those include a potentially greater alleviation of cognitive deficits and the existence of calming effects that may reduce aggression and hostility, such as during acute exacerbations of schizophrenia.
Cochrane reviews (Asmal, 2013 and Suttajit, 2013) have found it’s similar in efficacy to second-gen drugs like clozapine, aripiprazole, and ziprasidone. While olanzapine, risperidone, and paliperidone could be superior.
Reviews indicate it has a lower movement disorder potential than first-gen drugs and some second-gen drugs (e.g. paliperidone, risperidone, and olanzapine). It’s metabolic effects are considered “intermediate” in the second-gen class.
Along with treating the core psychotic symptoms of schizophrenia, quetiapine appears to alleviate mood symptoms, so it can assist with reducing depression.
Doses over 300 mg appear ideal for this condition. Some patients end up even higher than the recommended maximum of 750 mg/d. It’s not clear how high the dose range should go.
Cognition
Quetiapine seems to improve attention, motor and visuomotor skills, and executive function. This impact on cognition may be superior to what’s offered by first-gen antipsychotics.
Some evidence suggests unmedicated patients retain better verbal fluency and memory performance, though these results are challengeable. The sedative effects of quetiapine could also reduce cognitive performance in some patients.
Sexual dysfunction
Sexual dysfunction is a major problem with some antipsychotics, contributing to reduced compliance and a lower quality of life. It appears to be a relatively minor issue with quetiapine and when it exists, the symptoms aren’t especially severe.
Data from AstraZeneca found the rate of impotence, abnormal ejaculation, and amenorrhea (absence of menstruation) was under 0.1%.
Priapism (an erection lasting hours) has been reported in a few cases. It’s a very rare response.
Quetiapine’s improved profile in this area may be partly the result of comparatively low effects on prolactin. Most patients don’t have a substantial rise in prolactin and those switching from other antipsychotics can see a normalization of their levels. (Stevens, 2005) found prolactin was above the upper limit of normal in 68% of males receiving risperidone, compared to 20% receiving quetiapine.
(Kelly, 2005) – A double-blind 12-week trial showing orgasm quality and ability were significantly better with quetiapine compared to fluphenazine and risperidone.
The greatest issue may be a reduction in libido. (Atmaca, 2005) found diminished libido in a third of males and males.
Bipolar Disorder
Bipolar disorder is a psychiatric condition featuring marked fluctuations in mood, including episodes of depression and mania. It’s split into two core categories in the DSM-5: Bipolar 1 and Bipolar 2.
Bipolar 1 involves depression accompanied by full manic episodes, while Bipolar 2 involves depression with at least one hypomanic episode.
Quetiapine is effective in all phases of the disorder, including in manic episodes, mixed episodes, and bipolar depression. Efficacy has also been shown for maintenance on its own and as an add-on.
Compared to other antipsychotics, quetiapine has some of the most evidence supporting its use. It was one of the first antipsychotics approved by the FDA as a first-line treatment for both manic and depressive phases.
Acute states
When antipsychotics are added to classic mood stabilizers, there may be a faster onset of action and greater control of agitation and psychotic behavior during mania.
There are some case reports of it inducing manic and hypomanic states. This response is atypical and can often be addressed with dose reduction or treatment cessation.
It’s recommended to use a classic mood stabilizer, namely lithium or valproate, with an antipsychotic like quetiapine for severe manic episodes. Milder cases can be treated with a single drug.
Studies in bipolar mania have shown it may have a similar efficacy to lithium for acute mania (Bowden, 2005 and McIntyre, 2005). Manic or hypomanic patients receiving lithium or valproate seem to do better if they also receive quetiapine (Bahk, 2004).
(Smith, 2007) – A review of 13 trials with 3,089 patients. It compared the effects of carbamazepine, haloperidol, lithium, olanzapine, quetiapine, risperidone, valproate, and aripiprazole in acute bipolar mania. Only 2 studies evaluated quetiapine and because the response rate varied from below placebo to 137% greater than placebo , it was found to be the only drug lacking a significant response difference vs. placebo.
Quetiapine can alleviate, even on its own, bipolar depression (Calabrese, 2004). It seems to reduce core depressive symptoms, suicidal ideation, and anxiety.
A large review of its use for acute bipolar depression (Suttajit, 2014) used data from 11 studies and 3,488 patients. All of the trials were 1 to 12 weeks long. They compared quetiapine to placebo, SSRIs, lithium, and non-drug IPSRT therapy. Quetiapine was as or more effective than SSRIs, lithium, and non-drug therapy. It’s worth nothing these studies were sponsored by the maker of quetiapine, AstraZeneca.
Maintenance treatment
The only FDA-approved way of using quetiapine for maintenance is if it’s combined with a first-line treatment (valproate or lithium). Limited studies (often small sample sizes and open-label designs) suggest it could work on its own (Altamura, 2003).
Rapid-cycling
Patients with rapid-cycling bipolar disorder often have poor or no response to classic mood stabilizers. Quetiapine appears useful in those situations, particularly when added to typical treatments (Vieta, 2002).
BDNF
Some evidence points to impaired BDNF levels (Brain-Derived Neurotrophic Factor) in bipolar disorder. Levels might be low in acute mood episodes, while returning to normal during a positive non-manic mood state (euthymia). If someone goes through multiple episodes, BDNF levels could remain low (Kauer-Sant’Anna, 2009). There is opposing evidence, however. (Magalhaes, 2012) showed young people with bipolar disorder have normal BDNF levels.
As this is an active area of research, more studies have tried to see if bipolar is linked to impaired BDNF and if treatment normalizes that issue.
(Grande, 2012) – Open-label trial with 25 patients. It found BDNF levels increased for those initially in a depressed state, but they significantly declined if people started quetiapine while in a manic or mixed state.
Insomnia
It’s not unheard of for antipsychotics or antidepressants to be used for insomnia. Of those drugs, quetiapine has received more attention than most. It’s one of the most common conditions treated by quetiapine even though it’s not approved for that use and formal evidence is lacking.
The only time it’s officially recommended is if insomnia is secondary to a comorbid disorder like psychosis or depression. Tolerance may build quickly, though a small open-label pilot study (Wiegand, 2008) found it offered improved objective and subjective sleep measures in primary insomnia lasting up to 6 weeks. The starting dose was 25 mg. 7 participants ended up using 50 mg and 1 participant moved up to 75 mg.
(Cohrs, 2004) – The sleep promoting effects were evaluated in 18 healthy males. It was found to significantly increase sleep time, sleep efficiency, and percentage of time spent in Stage 2. REM was reduced. Participants self-reported better sleep when receiving 25 or 100 mg and they said they felt more refreshed.
More evidence exists supporting alternative substances and non-drug options.
Some people only use it occasionally, in which case quetiapine appears quite effective. But it can cause drowsiness the following day, even with the low 25-100 mg doses that are used.
Depression
It’s often used as an antidepressant even in patients suffering from unipolar rather than bipolar depression. Quetiapine is only approved as an add-on treatment for depression, specifically in its extended-release form.
Studies have reliably found it can reduce depression in bipolar disorder and schizophrenia. Some evidence exists supporting antidepressant properties, either on its own or with other drugs, in major depression.
In 2009, an FDA advisory committee recommended against approval for depression and anxiety primarily due to concerns about metabolic issues, sudden cardiac death, and movement disorders. The committee recognized it may have efficacy, but concerns over side effects outweighed the perceived benefits.
(Weisler, 2012) – Analysis of two 6-week, double-blind, controlled studies. 968 patients with major depression were randomized to quetiapine XR 150 mg, 300 mg, or placebo. The quetiapine groups experienced a significantly higher response rate and and total depression symptom reduction, albeit with small effect sizes. The reductions were significant from Week 1 onward. Only 300 mg led to a significantly higher remission rate.
Anxiety
Though it’s not approved for anxiety, it’s often used for that purpose, especially for generalized anxiety disorder (GAD). Like with depression, studies have shown efficacy, but the effect sizes are often pretty small compared to placebo (Stein, 2011). It may also be used as an add-on drug.
(Katzman, 2010) – Longer-term randomized, controlled trial of quetiapine XR for GAD. 432 patients were stabilized on open-label quetiapine and then randomized to continue quetiapine or placebo. Those receiving quetiapine had a significantly greater reduction in anxiety and fewer (10.2% vs. 38.9%) experienced a recurrence of anxiety symptoms.
(Kreys, 2015) – A review of the literature found quetiapine XR was effective on its own or with other treatments. Efficacy appeared by Week 1 of treatment.
Delirium
No antipsychotic is specifically FDA-approved for delirium in hospital settings. Haloperidol has historically been used more than any other.
Some studies (Maneeton, 2013 and Devlin, 2010) show quetiapine is also effective for this purpose and it’s largely equal in efficacy to haloperidol.
Substance use
There’s interest in using quetiapine as a treatment for various substance use disorders, most often alcohol. A limited set of evidence stemming from small trials, case reports, and chart reviews, suggests it could reduce craving and alcohol intake. This could conceivably be from a direct effect on craving, potentiation of unpleasant sedative effects, blunting of positive effects, or improved anxiety and sleep (thereby reducing interest in using alcohol for self-medication).
(Martinotti, 2008) – An open-label trial showing positive effects in people with alcohol use disorder and another psychiatric condition, like bipolar disorder or schizoaffective disorder.
(Kennedy, 2008) – An open trial for cocaine use disorder in non-psychotic individuals. 22 participants entered the trial; only 14 finished. Craving and use declined significantly in the ITT group. The effect size (1.23) was large.
(Cooper, 2012) – This lab study exposed daily cannabis users to cannabis use and withdrawal conditions while giving them 200 mg/d of quetiapine or placebo. Compared with placebo, quetiapine improved sleep quality, increased caloric intake, and decreased weight loss during the withdrawal period. But it also increased cannabis craving and cannabis self-administration when people were given active cannabis again, so it was deemed of little use for cannabis use disorder.
Borderline personality disorder
Antipsychotics, including quetiapine, may reduce impulsivity, aggression, depression, and anxiety associated with borderline personality disorder. Only a limited amount of evidence exists, so a conclusion can’t be reached.
(Eynde, 2008) – A 12-week open trial with 41 patients (32 of whom completed). Patients had significant reductions in impulsivity, hostility, and aggression. And they had significantly better overall functioning. The mean end dose was 412.5 mg.
Psychosis or agitation associated with Parkinson’s or dementia
The evidence so far indicates it’s not an effective treatment for psychotic symptoms or agitation associated with neurodegeneration.
(Rabey, 2007) – 12-week, double-blind, controlled study. 58 patients with Parkinson’s and psychotic symptoms were randomized to quetiapine (n=30) or placebo (n=28). The patients began the study with severe psychotic symptoms. There was a high dropout rate (50% for quetiapine and 39% for placebo). The average dose was 119 mg. Quetiapine failed to show any significant benefit for psychotic symptoms.
It wasn’t significantly better than placebo for agitation or psychosis associated with dementia or parkinsonism (Kurlan, 2007).
It wasn’t effective at reducing agitation compared to placebo in a double-blind, controlled study in Alzheimer’s disease. Quetiapine patients also had greater cognitive decline (Ballard, 2005).
Other medical uses
Non-medical
It doesn’t have any notable recreational effects, but some people take it for sedation, sleep-induction, and possibly anxiolysis outside of medical settings. There are reports of people “abusing” it, often in prison/institutionalized settings and in people with a history of psychiatric disorders or substance abuse.
There are reports of people taking it to come down from stimulants, to potentiate other depressants, and to alleviate withdrawal symptoms from various other drugs.
Chemistry & Pharmacology
Chemistry
Quetiapine is a dibenzothiazepine derivative with structural similarities to clozapine. They contain a similar tricyclic heterocycle structure.
One of the nitrogen heteroatoms within clozapine’s dibenzodiazepine ring has been replaced with a sulfur in quetiapine, yielding a dibenzothiazepine ring.
Both drugs also have a piperazine ring substituted at the 4-position of their main tricyclic heterocycle.
Pharmacology
Quetiapine affinity (Jensen, 2008)
- D2 – 245 nM
- 5-HT2A – 100 nM
- 5-HT2C – 2502 nM
- 5-HT1A – 432 nM
- 5-HT7 – 307 nM
- H1 – 11 nM
- a1-adrenergic – 22 nM
- a2-adrenergic – 3630 nM
- NET – Over 10,000 nM
Norquetiapine affinity (Jensen, 2008)
- D2 – 196 nM
- 5-HT2A – 48 nM
- 5-HT2C – 107 nM
- 5-HT1A – 45 nM
- 5-HT7 – 76 nM
- H1 – 3.5 nM
- a1-adrenergic – 144 nM
- a2-adrenergic – 237 nM
- NET – 58 nM
Unlike first-gen antipsychotics, its affinity for dopamine D2 receptors is lower than its affinity for other receptors. It has a higher affinity at 5-HT2A, histamine H1, and a1-adrenergic. It’s an antagonist at all of those sites, while it’s a moderate affinity agonist at 5-HT1A.
Its low affinity for D2 and its fast dissociation rate is thought to be the reason movement disorders are less of an issue. Occupancy at therapeutic doses is only around 30% (Gefvert, 2001), whereas first-gen antipsychotics typically have an occupancy of 60-75%. That occupancy is substantially lower than for 5-HT2A (around 75%). Because of its lower D2 binding, there may also be a faster return of psychotic symptoms after each dose.
Other relevant factors in its reduced potential for movement disorders are its short half-life at D2 receptors (~10 hours) and a potential preference for mesolimbic and mesocortical dopamine pathways over the nigrostriatal pathways that’d lead to EPS (Goldstein, 1999).
Its pharmacological profile varies by dose. At low doses (~25 mg), it’s mainly an H1 antagonist. Moderate doses (50-100+ mg) incorporate greater serotonin receptor antagonism. High doses (300+ mg) recruit D2 antagonism.
Norquetiapine
Norquetiapine reaches around 1/3 the Cmax of quetiapine. Enough seems to be present for its pharmacology to contribute. As such, its actions at the following targets may be important: 5-HT2A (antagonist), 5-HT1A (agonist), 5-HT2C (antagonist), D2 (antagonist), NET (inhibitor).
Its pharmacology overlaps significantly with that of other antipsychotics, antidepressants, and anxiolytics. Two binding sites in particular, 5-HT1A and NET inhibition, are popular targets for anxiolytic and antidepressant drugs. Therefore it seems possible norquetiapine is contributing to that aspect of quetiapine’s activity.
Norquetiapine’s impact at NET is distinct from that of other antipsychotics, which have no or little affinity (Cross, 2016). That aspect of the drug is more akin to what’s seen with antidepressants like duloxetine and imipramine.
It’s been found to exhibit antidepressant and anxiolytic effects in animals. At least some of the anxiolysis is blocked by a 5-HT1A antagonist.
Actions through non-neurotransmitter systems
Some studies have found anti-inflammatory and antioxidant effects with quetiapine. It inhibits nitric oxide generation and TNF-a release from IFN-y-activated microglia in vitro (Bian, 2008).
Along with potentially increasing antioxidant status through a rise in superoxide dismutase (SOD) (Miljevic, 2013), it seems to have direct antioxidant potential. (Xu, 2008) found it directly functioned as a ROS scavenger in an in vitro Alzheimer’s disease model. It was shown to reduce hydroxyl radical concentration in a concentration-dependent manner.
It’s been shown to increase SOD1 (superoxide dismutase) activity in human erythrocytes in vitro and it upregulated SOD1 gene expression (Bai, 2002). But there is a conflicting result in (Schmidt, 2009) showing a drop in mRNA expression of genes encoding for key antioxidant enzymes, including SOD, mnSOD, glutathione peroxidase, and catalase. In that study the effects were seen with 300 uM.
Neuroprotection
It has neuroprotective properties in vitro and in animals. The protective effects are active against models of Alzheimer’s disease (Xu, 2008), stress-induced impairments (Xu, 2006), ischemia-induced degeneration (Yan, 2007), and methamphetamine toxicity (He, 2006).
Most studies evaluating these kinds of effects have been positive, though chronic quetiapine (5 or 10 mg/kg SC) in rats failed to protect against phencyclidine-induced increased impulsivity and disrupted attention (Amitai, 2008).
Its ability to block a stress-induced decline in cellular proliferation and BDNF expression may be relevant to psychiatric conditions, including depression and anxiety. Supporting BDNF and cellular proliferation are two mechanisms seen with some antidepressants.
Genetic effects
Sensorimotor gating
Sensorimotor gating can be assessed by the acoustic startle reflex in humans and animals. Prepulse inhibition (PPI) is used to see how much a weak prepulse inhibits the brain’s response to a subsequent full “pulse.” PPI appears to be impaired in some psychiatric conditions, including schizophrenia.
Animal studies have shown quetiapine reverses the PPI-disrupting effects of lesions (Shoemaker, 2003) and the PPI-disrupting effects of knocking out the dopamine transporter (DAT) (Powell, 2008).
fMRI
Treatment with quetiapine may restore greater cortical activity in patients with schizophrenia. That increase in activity could underlie some of the cognitive and emotional benefits.
(Meisenzahl, 2006) – A trend towards improved working memory with quetiapine in patients with schizophrenia. This was accompanied by greater activation during the task in the prefrontal cortex.
Study with 12 healthy controls and 12 patients with schizophrenia. Their working memory performance and neural correlates were studied. The controls naturally had significantly greater activation in the left thalamus, basal ganglia, right DLPFC and in the bilateral VLPFC during a cognitive task. That increase in activation didn’t occur in patients. When quetiapine was used for 12 weeks (mean dose: 566.7 mg/d), an increase in activation was seen, with a significant rise in the left VLPFC during 2-back conditions.
(Stip, 2005) – Restoration of frontal activation in patients with schizophrenia. This was associated with improved affective symptoms, namely relief of blunted affect.
5.5 months of treatment (mean dose: 529 mg/d) in 12 participants led to significant reductions in blunted affect. Those reductions were accompanied by increased prefrontal cortex activation when the patients were shown emotional film excerpts.
(Jones, 2004) – Greater frontal cortex activation during a verbal fluency task and temporal cortex activation during auditory stimulation in patients with schizophrenia given quetiapine.
This study looked at 7 drug-naive patients, 8 quetiapine patients, and 8 healthy controls. In both situations, quetiapine patients (mean dose: 364 mg/d for 5.7 months) showed significantly greater activation than drug-naive patients. This was the case in the left inferior frontal cortex during the verbal fluency task. In the auditory stimulation condition, greater activation was observed in the superior temporal gyrus.
(Klasen, 2013) – Modulates functional connectivity in brain aggression networks in healthy people.
13 healthy males were tested in a double-blind, cross-over manner while playing a violent video game meant to induce “aggression.” Functional connectivity between the amygdala and dorsal anterior cingulate cortex (dACC) in the quetiapine condition. Functional connectivity also increased with the DLPFC. Whereas violence-specific connectivity declined between the amygdala and right orbitofrontal cortex (OFC).
Pharmacokinetics
Tmax: 1 – 2 hours
Half-life: 7 hours
Protein binding: 83%
Excretion: 73% in urine and 21% in feces
Interactions
Phenytoin is a potent inducer of CYP3A4, so higher doses of quetiapine may be needed.
(DeVane, 2001) – Neither risperidone nor haloperidol impacted its kinetics, but thioridazine produced decreased in the AUC (-40%), Cmax (-22%), and Cmin (-31%). This occurred by an unknown mechanism.
Ketoconazole produced a 4.03x greater Cmax.
Polymorphisms
(Weide, 2014) – The impact of CYP3A4 polymorphisms was studied in 238 adult patients. Carriers of the CYP3A4*22 allele had a 2.5-fold higher serum concentration compared to wild-type patients receiving a similar dose. The dose-corrected serum concentration was 67% higher in carriers.
The kinetics of quetiapine appear unaffected by dose, ethnicity, gender, and weight. Bioavailability of the IR formulation isn’t affected by food. In elderly patients the target dose showed be lowered because mean plasma clearance is reduced 30-50%.
Its primary metabolic route is via CYP3A4. The most abundant metabolites appear to be quetiapine carboxylic acid and quetiapine sulfoxide. Together they account for ~30% of the quetiapine dose.
At least one active metabolite, norquetiapine, seems relevant. Another (7-hydroxyquetiapine) is also produced, but only very small concentrations are reached.
History
1985
Quetiapine was developed by AstraZeneca.
1997
Quetiapine was approved for use in the US by the FDA in September 1997. It entered the market under the name “Seroquel.”
2006
It was by this point approved for use and available in over 80 countries worldwide.
2007
Three second-gen antipsychotics (olanzapine, risperidone, and quetiapine) were among the 10 top-selling drugs worldwide, with a combined sales volume of $14.5 billion.
2008
Quetiapine sales reached nearly $4.5 billion.
2009
According to the Associated Press, the Pentagon and Department of Veterans Affairs spent $8.6 million and $125.4 million on the drug, respectively.
2009
In April 2009, the FDA’s Psychopharmacologic Drugs Advisory Committee was asked to advise the FDA on whether quetiapine should be approved for anxiety and depression. Dr. Thomas Laughren (director of the FDA’s Division of Psychiatric Products) noted that although AstraZeneca’s data showed efficacy, the agency was concerned about more widespread use due to the metabolic, movement disorder, and cardiac risks.
Dr. Wayne Goodman (the panel’s acting chair and director of adult translational research at the NIMH) said the panel found quetiapine acceptably safe as an add-on for depression (Vote: 6 to 3). This decision wasn’t considered precedent-setting because the agency previously approved the antipsychotic aripiprazole for that use.
Due to the side effect potential, it wasn’t recommended the drug be approved for depression or anxiety as a single therapy.
2010
AstraZeneca paid a $520 million settlement due to improper marketing of quetiapine and improper handling of some clinical trials. Because of multiple lawsuits filed against the drug maker, some of the company’s internal activities were revealed.
Internal emails reveal it buried and misrepresented studies showing a lack of superior efficacy compared to first-gen drugs. It did the same with studies showing a notable risk of obesity and weight gain.
The emails show employees clearly knew the results were damaging to the drug’s potential, but they kept sharing inaccurate information and hiding useful data from physicians and the public. AstraZeneca also fought the FDA when the agency requested it inform doctors about a greater than previously suggested risk of diabetes.
In one case, the company learned of clinical data pointing to significant weight gain potential. Rather than discuss that potential side effect, it presented data showing weight loss at an American Psychiatric Association conference.
2010
(Monasterio, 2011) – Survey of off-label second-gen antipsychotic prescribing by psychiatrists in Christchurch, New Zealand. Quetiapine was the preferred drug in 94% of cases and 59% of respondents prescribed it off-label every week in cases with little scientific evidence of safety and efficacy.
2000s to 2010s
Data from PHARMAC (pharmaceutical funding agency in New Zealand) was used to look at prescribing trends over 10 years. There was a 700% increase in quetiapine prescribing and a shift from specialists to general practitioners. GPs prescribed 2.5x more quetiapine than specialists in 2011, whereas specialists prescribed 4x more than GPs in 2002.
GPs were also more likely to prescribe low-dose quetiapine.
In 2011, 36% of GP prescribing and 19% of specialist prescribing was for under 100 mg/d.
2012
The IR formulation became available as a generic drug in the US in March 2012.
2013
An IMS Health report showed quetiapine was the most frequently prescribed second-gen antipsychotic in the US with over 14 million dispensed prescriptions in 2013.
2017
AstraZeneca was still dealing with whistleblower lawsuits over misrepresentation of data and off-label marketing. Those legal actions reveal it may have hidden risks about combining quetiapine with other medications capable of QT/QTc prolongation, which raises the risk of sudden cardiac death from fatal arrhythmias.
Legal Status (as of November 2017)
United States
Prescription-only
Other countries
It’s also prescription-only in Australia, Canada, and the UK.
Safety
Overdose
It has a favorable overdose profile, with very few deaths coming from direct toxicity. Though it does come with some potential for coma, respiratory depression, rhabdomyolysis, and cardiovascular issues (like a prolonged QT interval and Torsades de pointes), the outcome of overdoses is rarely death.
Some of the most common symptoms are increased heart rate (tachycardia) and drowsiness, including to the point of unconsciousness. Some other potential symptoms include hypotension, delirium, and seizures.
Because there are quite a few cases where mechanical ventilation or other significant interventions have been required, more deaths would exist if not for medical treatment.
Movement disorders and neuroleptic malignant syndrome
Some research has found quetiapine causes no increased risk of EPS, even in vulnerable groups like the elderly, adolescents, or patients with organic brain disorders.
Because other studies have found a somewhat elevated rate of EPS, it doesn’t appear entirely risk-free, but it less problematic than first-gen antipsychotics. There have been rare reports of neuroleptic malignant syndrome (Kobayashi, 2006) and tardive dyskinesia as well. Again, those issues are less common than with first-gen antipsychotics.
Metabolic
Quetiapine has a verified connection to weight gain and it may raise your risk of obesity, diabetes, and hyperlipidemia. Typically the effects are limited to relatively moderate weight gain, with some studies failing to even show an increase in weight. But a minority of patients will experience more problematic effects, raising their risk of various disorders in the long-term.
Significant cholesterol changes usually aren’t reported. Increases in fasting triglycerides and glucose are more common. A triglyceride level over 200 mg/dl may be upwards of 3x more common with quetiapine than with placebo (Stein, 2011).
The bulk of the evidence points to an average weight increase of 1 – 3 kg (2 – 7 lbs) over a period of a few months. Based on a limited number of long-term studies, it doesn’t seem to progressively increase weight indefinitely.
(Rummel-Kluge, 2010) – A head-to-head comparison of the metabolic side effects of second-gen antipsychotics in the treatment of schizophrenia. This review yielded 45 studies, 11 of which included quetiapine. Overall, olanzapine and clozapine were worst for weight, cholesterol, and glucose changes. Quetiapine, risperidone, and sertindole had intermediate effects,. Aripiprazole and amisulpride had intermediate or low elevations. Ziprasidone had the lowest impact.
Endocrine
It has minimal impact on prolactin levels, making it superior in that regard to some antipsychotics. The lack of impact on prolactin could be due to reduced dopamine antagonism in the tuberohypophyseal pathway relative to other antipsychotics.
(de Borja, 2005) – In healthy volunteers it produced an acute rise in prolactin and a decline in cortisol with 150 mg. There was also a significant increase in growth hormone secretion later on.
(Kelly, 2005) – Based on data from 38 patients, quetiapine had no influence on thyroid function.
Seizures
(Dev, 2000) – A clinical trial with 3,700 patients didn’t show an increased rate of seizures.
Pregnancy
The available data doesn’t indicate a connection to major malformations during pregnancy.
Hematological
(Croarkin, 2001) – 1% of patients develop leukopenia (reduced white blood cell count; raises infection risk). This resolves with discontinuation of the drug.
Psychological
Occasional reports of drug-induced mania, hypomania, or depression exist.
Hepatic (liver)
Quetiapine is typically considered safe in regards to its impact on liver function, but there are reports of elevated liver enzymes, elevated bilirubin, and rarely hepatotoxicity.
Around a quarter of patients (Atasoy, 2007) could have an asymptomatic elevation in liver enzymes after starting the drug. That elevation appears to decline over time.
Cardiovascular
Multiple studies have shown a connection between first-gen antipsychotics and sudden cardiac death. It was long believed second-gen drugs like quetiapine came with a substantially lower risk, but that may not be the case. Both classes have been shown to prolong the QT interval and using antipsychotics is a risk factor for sudden death.
The risks are even greater if you combine quetiapine with another drug capable of QT prolongation.
(Ray, 2009) – A major epidemiologial study showed an increased risk of sudden cardiac death with second-gen antipsychotics. Using data on 93,300 antipsychotic users and 186,000 controls, current users of both first- and second-gen antipsychotics had a higher risk of sudden cardiac death. Former users didn’t have a higher risk than the non-using population. Differences were found within the classes. For second-gen drugs, quetiapine’s relative risk was half that of clozapine’s. The risk with quetiapine is dose-dependent, with low doses (under 75 mg/d) showing minimal increase in risk and high doses showing an increase akin to that of first-gen drugs.
Because there are cardiovascular risks, limiting use in more vulnerable populations (e.g. the elderly) could make sense.
Dependence
Physical dependence does exist with quetiapine and withdrawal symptoms are possible when use stops. Those symptoms are worst with abrupt discontinuation, so tapering is recommended.
The symptoms can include sweating (diaphoresis), increased blood pressure, vomiting, lightheadedness, nausea, nervousness, insomnia, anxiety, and increased heart rate (tachycardia).
A return of psychotic symptoms is also possible.
Risky combos (list may not be complete)
- Other depressants, including alcohol.
- Drugs that can prolong the QT interval.
References
A., H., D., K., & S., G. (2010). Smoking quetiapine: A “maq ball”? Primary Psychiatry. Retrieved from http://mbldownloads.com/0910PP_Haridas.pdf%5Cnhttp://ovidsp.ovid.com/ovidweb.cgi?T=JS&PAGE=reference&D=emed9&NEWS=N&AN=2010661387
Adler, C. M., DelBello, M. P., Weber, W. A., Jarvis, K. B., Welge, J., Chu, W.-J., … Strakowski, S. M. (2013). Neurochemical Effects of Quetiapine in Patients With Bipolar Mania. Journal of Clinical Psychopharmacology, 33(4), 528–532. https://doi.org/10.1097/JCP.0b013e3182905b77
Alexander, J. (2010). Delirium as a symptom of tricyclic. World Health, 2010. https://doi.org/doi:10.3109/00048674.2010.497477
Amitai, N., & Markou, A. (2009). Increased impulsivity and disrupted attention induced by repeated phencyclidine are not attenuated by chronic quetiapine treatment. Pharmacology Biochemistry and Behavior, 93(3), 248–257. https://doi.org/10.1016/j.pbb.2008.08.025
Anderson, S. L., & Vande Griend, J. P. (2014). Quetiapine for insomnia: A review of the literature. American Journal of Health-System Pharmacy. https://doi.org/10.2146/ajhp130221
Antipsychotic Agents and Sudden Cardiac Death —How Should We Manage the Risk? (2008), 1–3. Retrieved from http://www.nejm.org.proxy.bib.uottawa.ca/doi/pdf/10.1056/NEJMe0809417%5Cnpapers2://publication/uuid/8DDDBA94-2FCD-4B21-8D40-DBBDD427138F
Arvanitis, L. A. (1997). OR OINA Multiple Fixed Doses of “Seroquel” Patients with Acute A Comparison with Exacerbation of Haloperidol and (Quetiapine) in Schizophrenia: Placebo Miller, and the Seroquel Trial 13 Study Group. BIOL PSYCHIATRY, 4242.
Asmal, L., Flegar, S. J., Wang, J., Rummel-Kluge, C., Komossa, K., & Leucht, S. (2013). Quetiapine versus other atypical antipsychotics for schizophrenia. In Cochrane Database of Systematic Reviews. https://doi.org/10.1002/14651858.CD006625.pub3
Atasoy, N., Erdogan, A., Yalug, I., Ozturk, U., Konuk, N., Atik, L., & Ustundag, Y. (2007). A review of liver function tests during treatment with atypical antipsychotic drugs: A chart review study. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 31(6), 1255–1260. https://doi.org/10.1016/j.pnpbp.2007.05.005
Bakken, G. V., Rudberg, I., Christensen, H., Molden, E., Refsum, H., & Hermann, M. (2009). Metabolism of quetiapine by CYP3A4 and CYP3A5 in presence or absence of cytochrome B5. Drug Metabolism and Disposition, 37(2), 254–258. https://doi.org/10.1124/dmd.108.023291
Ballard, C. (2005). Quetiapine and rivastigmine and cognitive decline in Alzheimer’s disease: randomised double blind placebo controlled trial. BMJ, 330(7496), 874–0. https://doi.org/10.1136/bmj.38369.459988.8F
Barzman, D. H., DelBello, M. P., Adler, C. M., Stanford, K. E., & Strakowski, S. M. (2006). The Efficacy and Tolerability of Quetiapine versus Divalproex for the Treatment of Impulsivity and Reactive Aggression in Adolescents with Co-Occurring Bipolar Disorder and Disruptive Behavior Disorder(s). Journal of Child and Adolescent Psychopharmacology, 16(6), 665–670. https://doi.org/10.1089/cap.2006.16.665
Bowden, C., & Bowden, V. (1992). Annals of Clinical Psychiatry. JAMA: The Journal of the American Medical Association. https://doi.org/10.1001/jama.1992.03480050135044
Brown, E. S., Nejtek, V. A., Perantie, D. C., & Bobadilla, L. (2002). Quetiapine in bipolar disorder and cocaine dependence. Bipolar Disorders, 4(6), 406–411. https://doi.org/10.1034/j.1399-5618.2002.02229.x
Buckley, P. F., Goldstein, J. M., & Emsley, R. A. (2004). Efficacy and tolerability of quetiapine in poorly responsive, chronic schizophrenia. Schizophrenia Research, 66(2–3), 143–150. https://doi.org/10.1016/j.schres.2003.06.001
Byers, M. G., Allison, K. M., Wendel, C. S., & Lee, J. K. (2010). Prazosin Versus Quetiapine for Nighttime Posttraumatic Stress Disorder Symptoms in Veterans. Journal of Clinical Psychopharmacology, 30(3), 225–229. https://doi.org/10.1097/JCP.0b013e3181dac52f
Calabrese, J. R., Keck, P. E., Macfadden, W., Minkwitz, M., Ketter, T. A., Weisler, R. H., … Group, T. (2005). A randomized, double-blind, placebo-controlled trial of quetiapine in the treatment of bipolar I or {II} depression. The American Journal of Psychiatry, 162(7), 1351–1360. https://doi.org/10.1176/appi.ajp.162.7.1351
Calabrese, J. R., Keck, P. E., Macfadden, W., Minkwitz, M., Ketter, T. A., Weisler, R. H., … Jamie Mullen, M. (2005). A Randomized, Double-Blind, Placebo-Controlled Trial of Quetiapine in the Treatment of Bipolar I or II Depression The BOLDER Study Group. Am J Psychiatry, 1627. Retrieved from http://ajp.psychiatryonline.org
Caniato, R. N., Gundabawady, A., Baune, B. T., & Alvarenga, M. E. (2009). Malingered psychotic symptoms and quetiapine abuse in a forensic setting. Journal of Forensic Psychiatry and Psychology, 20(6), 928–935. https://doi.org/10.1080/14789940903174196
Capuano, A., Ruggiero, S., Vestini, F., Ianniello, B., Rafaniello, C., Rossi, F., & Mucci, A. (2011). Survival from coma induced by an intentional 36-g overdose of extended-release quetiapine. Drug and Chemical Toxicology, 34(4), 475–477. https://doi.org/10.3109/01480545.2011.564178
Chang, K., DelBello, M., Chu, W.-J., Garrett, A., Kelley, R., Mills, N., … Strakowski, S. M. (2012). Neurometabolite Effects of Response to Quetiapine and Placebo in Adolescents with Bipolar Depression. Journal of Child and Adolescent Psychopharmacology, 22(4), 261–268. https://doi.org/10.1089/cap.2011.0153
Chernoloz, O., El Mansari, M., & Blier, P. (2012). Effects of sustained administration of quetiapine alone and in combination with a serotonin reuptake inhibitor on norepinephrine and serotonin transmission. Neuropsychopharmacology, 37(7), 1717–1728. https://doi.org/10.1038/npp.2012.18
Coe, H. V., & Hong, I. S. (2012). Safety of Low Doses of Quetiapine When Used for Insomnia. Annals of Pharmacotherapy, 46, 718–722. https://doi.org/10.1345/aph.1Q697
Cohrs, S., Rodenbeck, A., Guan, Z., Pohlmann, K., Jordan, W., Meier, A., & Rüther, E. (2004). Sleep-promoting properties of quetiapine in healthy subjects. Psychopharmacology, 174, 421–429. https://doi.org/10.1007/s00213-003-1759-5
Cooper, Z. D., Foltin, R. W., Hart, C. L., Vosburg, S. K., Comer, S. D., & Haney, M. (2013). A human laboratory study investigating the effects of quetiapine on marijuana withdrawal and relapse in daily marijuana smokers. Addiction Biology, 18(6), 993–1002. https://doi.org/10.1111/j.1369-1600.2012.00461.x
Cross, A. J., Widzowski, D., Maciag, C., Zacco, A., Hudzik, T., Liu, J., … Wood, M. W. (2016). Quetiapine and its metabolite norquetiapine: Translation from in vitro pharmacology to in vivo efficacy in rodent models. British Journal of Pharmacology, 173(1), 155–166. https://doi.org/10.1111/bph.13346
Cubała, W. J., & Springer, J. (2014). Quetiapine abuse and dependence in psychiatric patients: a systematic review of 25 case reports in the literature. Journal of Substance Use, 19(5), 388–393. https://doi.org/10.3109/14659891.2013.810309
DeVane, D. C. L., & Nemeroff, C. B. (2001). Clinical Pharmacokinetics of Quetiapine. Clinical Pharmacokinetics, 40(7), 509–522. https://doi.org/10.2165/00003088-200140070-00003
Devarajan, S., Ali, J., & Dursun, S. M. (2006). Quetiapine plus SSRI in treatment-resistant depression: Possible mechanisms [2]. Psychopharmacology. https://doi.org/10.1007/s00213-006-0314-6
Devlin, J. W., Roberts, R. J., Fong, J. J., Skrobik, Y., Riker, R. R., Hill, N. S., … Garpestad, E. (2010). Efficacy and safety of quetiapine in critically ill patients with delirium: A prospective, multicenter, randomized, double-blind, placebo-controlled pilot study*. Critical Care Medicine, 38(2), 419–427. https://doi.org/10.1097/CCM.0b013e3181b9e302
Dogu, O., Sevim, S., & Kaleagasi, H. S. (2003). Seizures associated with quetiapine treatment. Annals of Pharmacotherapy, 37(9), 1224–1227. https://doi.org/10.1345/aph.1C516
El-Khalili, N. (2012). Update on extended release quetiapine fumarate in schizophrenia and bipolar disorders. Neuropsychiatric Disease and Treatment. https://doi.org/10.2147/NDT.S14369
Eyer, F., Pfab, R., Felgenhauer, N., Strubel, T., Saugel, B., & Zilker, T. (2011). Clinical and analytical features of severe suicidal quetiapine overdoses – a retrospective cohort study. Clinical Toxicology, 49(9), 846–853. https://doi.org/10.3109/15563650.2011.624100
Fernandes, P. P., & Marcil, W. A. (2002). Death associated with quetiapine overdose [5]. American Journal of Psychiatry. https://doi.org/10.1176/appi.ajp.159.12.2114
Findling, R. L., McNamara, N. K., Gracious, B. L., O’Riordan, M. A., Reed, M. D., Demeter, C., & Blumer, J. L. (2004). Quetiapine in nine youths with autistic disorder. JOURNAL OF CHILD AND ADOLESCENT PSYCHOPHARMACOLOGY, 14(2), 287–294. https://doi.org/10.1089/1044546041649129
Findling, R. L., Mcnamara, N. K., Gracious, B. L., O ’riordan, M. A., Reed, M. D., Demeter, C., & Blumer, J. L. (2004). Quetiapine in Nine Youths with Autistic Disorder. JOURNAL OF CHILD AND ADOLESCENT PSYCHOPHARMACOLOGY, 14(2).
FINDLING, R. L., REED, M. D., O’RIORDAN, M. A., DEMETER, C. A., STANSBREY, R. J., & MCNAMARA, N. K. (2006). Effectiveness, Safety, and Pharmacokinetics of Quetiapine in Aggressive Children With Conduct Disorder. Journal of the American Academy of Child & Adolescent Psychiatry, 45(7), 792–800. https://doi.org/10.1097/01.chi.0000219832.23849.31
Fisher, D. S., Handley, S. A., Flanagan, R. J., & Taylor, D. M. (2012). Plasma Concentrations of Quetiapine, N-Desalkylquetiapine, O-Desalkylquetiapine, 7-Hydroxyquetiapine, and Quetiapine Sulfoxide in Relation to Quetiapine Dose, Formulation, and Other Factors. Therapeutic Drug Monitoring, 1. https://doi.org/10.1097/FTD.0b013e3182603f62
Gefvert, O., Lundberg, T., Wieselgren, I.-M., Bergström, M., Långström, B., Wiesel, F.-A., & Lindström, L. (2001). D_2 and 5HT_2A receptor occupancy of different doses of quetiapine in schizophrenia: a PET study. European Neuropsychopharmacology, 11(2), 105–110. Retrieved from www.elsevier.com
Grande, I., Kapczinski, F., Stertz, L., Colpo, G. D., Kunz, M., Cereser, K. M., … Magalhaes, P. V. S. (2012). Peripheral brain-derived neurotrophic factor changes along treatment with extended release quetiapine during acute mood episodes: An open-label trial in drug-free patients with bipolar disorder. Journal of Psychiatric Research, 46(11), 1511–1514. https://doi.org/10.1016/j.jpsychires.2012.08.017
Gugger, J. J., & Cassagnol, M. (2008). Low-dose quetiapine is not a benign sedative-hypnotic agent. American Journal on Addictions. https://doi.org/10.1080/10550490802266185
Harmon, T. J., Benitez, J. G., Krenzelok, E. P., & Cortes-Belen, E. (1998). Loss of consciousness from acute quetiapine overdosage. Journal of Toxicology. Clinical Toxicology, 36(6), 599–602. https://doi.org/10.3109/15563659809028056
Hawkins, S. B., Bucklin, M., & Muzyk, A. J. (2013). Quetiapine for the treatment of delirium. Journal of Hospital Medicine. https://doi.org/10.1002/jhm.2019
He, J., Yang, Y., Yu, Y., Li, X., & Li, X. M. (2006). The effects of chronic administration of quetiapine on the methamphetamine-induced recognition memory impairment and dopaminergic terminal deficit in rats. Behavioural Brain Research, 172(1), 39–45. https://doi.org/10.1016/j.bbr.2006.04.009
Hilger, E., Barnas, C., & Kasper, S. (2003). Quetiapine in the Treatment of Borderline Personality Disorder. The World Journal of Biological Psychiatry, 4(1), 42–44. https://doi.org/10.3109/15622970309167910
Hunfeld, N. G., Westerman, E. M., Boswijk, D. J., de Haas, J. A., van Putten, M. J., & Touw, D. J. (2006). Quetiapine in overdosage: a clinical and pharmacokinetic analysis of 14 cases. Therapeutic Drug Monitoring, 28(2), 185–189. https://doi.org/10.1097/01.ftd.0000185770.44502.51\r00007691-200604000-00007 [pii]
Hutton, P., Morrison, A. P., Yung, A. R., Taylor, P. J., French, P., & Dunn, G. (2012). Effects of drop-out on efficacy estimates in five Cochrane reviews of popular antipsychotics for schizophrenia. Acta Psychiatrica Scandinavica. https://doi.org/10.1111/j.1600-0447.2012.01858.x
Jensen, N. H., Rodriguiz, R. M., Caron, M. G., Wetsel, W. C., Rothman, R. B., & Roth, B. L. (2008). N-Desalkylquetiapine, a potent norepinephrine reuptake inhibitor and partial 5-HT1A agonist, as a putative mediator of quetiapine’s antidepressant activity. Neuropsychopharmacology, 33(10), 2303–2312. https://doi.org/10.1038/sj.npp.1301646
Jones, H. M., Brammer, M. J., O’Toole, M., Taylor, T., Ohlsen, R. I., Brown, R. G., … Pilowsky, L. S. (2004). Cortical effects of quetiapine in first-episode schizophrenia: A preliminary functional magnetic resonance imaging study. Biological Psychiatry, 56(12), 938–942. https://doi.org/10.1016/j.biopsych.2004.08.006
kandasamym. (2015). Annals of Clinical Psychiatry. https://doi.org/10.3109/10401230490522061
Kasper, S., & Müller-Spahn, F. (2000). Review of quetiapine and its clinical applications in schizophrenia. Expert Opinion on Pharmacotherapy, 1(4), 783–801. https://doi.org/10.1517/14656566.1.4.783
Katzman, M. A., Brawman-Mintzer, O., Reyes, E. B., Olausson, B., Liu, S., & Eriksson, H. (2011). Extended release quetiapine fumarate (quetiapine XR) monotherapy as maintenance treatment for generalized anxiety disorder. International Clinical Psychopharmacology, 26(1), 11–24. https://doi.org/10.1097/YIC.0b013e32833e34d9
Kennedy, A., Wood, A. E., Saxon, A. J., Malte, C., Harvey, M., Jurik, J., … Tapp, A. (2008). Quetiapine for the Treatment of Cocaine Dependence. Journal of Clinical Psychopharmacology, 28(2), 221–224. https://doi.org/10.1097/JCP.0b013e318166f50d
Khan, A., Joyce, M., Atkinson, S., Eggens, I., Baldytcheva, I., & Eriksson, H. (2011). A Randomized, Double-Blind Study of Once-Daily Extended Release Quetiapine Fumarate (Quetiapine XR) Monotherapy in Patients With Generalized Anxiety Disorder. Journal of Clinical Psychopharmacology, 31(4), 418–428. https://doi.org/10.1097/JCP.0b013e318224864d
Kim, D. R., & Staab, J. R. (2005). Quetiapine discontinuation syndrome [1]. American Journal of Psychiatry. https://doi.org/162/5/1020 [pii]\r10.1176/appi.ajp.162.5.1020
Kivircik Akdede, B. B., Alptekin, K., Kitiş, A., Arkar, H., & Akvardar, Y. (2005a). Effects of quetiapine on cognitive functions in schizophrenia. Progress in Neuro-Psychopharmacology and Biological Psychiatry. https://doi.org/10.1016/j.pnpbp.2004.11.005
Kivircik Akdede, B. B., Alptekin, K., Kitiş, A., Arkar, H., & Akvardar, Y. (2005b). Effects of quetiapine on cognitive functions in schizophrenia. Progress in Neuro-Psychopharmacology and Biological Psychiatry. https://doi.org/10.1016/j.pnpbp.2004.11.005
Klein-Schwartz, W., Schwartz, E. K., & Anderson, B. D. (2014). Evaluation of Quetiapine Abuse and Misuse Reported to Poison Centers. Journal of Addiction Medicine, 8(3), 195–198. https://doi.org/10.1097/ADM.0000000000000020
Klein, L., Bangh, S., & Cole, J. (2017). Intentional Recreational Abuse of Quetiapine Compared to Other Second-generation Antipsychotics. Western Journal of Emergency Medicine, 18(2), 243–250. https://doi.org/10.5811/westjem.2016.10.32322
Kobayashi, A., Kawanishi, C., Matsumura, T., Kato, D., Furukawa, R., Kishida, I., & Hirayasu, Y. (2006). Quetiapine-induced neuroleptic malignant syndrome in dementia with Lewy bodies: A case report. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 30(6), 1170–1172. https://doi.org/10.1016/j.pnpbp.2006.04.001
Koopmann, A., Von Der Goltz, C., Hermann, D., & Kiefer, F. (2011). Propofol addiction initiated by anesthetic use. American Journal of Psychiatry. https://doi.org/10.1176/appi.ajp.2010.10091293
Kreys, T.-J. M., & Phan, S. V. (2015). A Literature Review of Quetiapine for Generalized Anxiety Disorder. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 35(2), 175–188. https://doi.org/10.1002/phar.1529
Kuehn, B. M. (2009). FDA panel issues mixed decision on quetiapine in depression and anxiety. JAMA: Journal of the American Medical Association, 301(20), 2081–2082. https://doi.org/10.1001/jama.2009.708
Kurlan, R., Cummings, J., Raman, R., & Thal, L. (n.d.). Quetiapine for agitation or psychosis in patients with dementia and parkinsonism.
L??pez-Mu??oz, F., & ??lamo, C. (2013). Active metabolites as antidepressant drugs: The role of norquetiapine in the mechanism of action of quetiapine in the treatment of mood disorders. Frontiers in Psychiatry. https://doi.org/10.3389/fpsyt.2013.00102
Langman, L. J., Kaliciak, H. A., & Carlyle, S. (2004). Fatal overdoses associated with quetiapine. Journal of Analytical Toxicology, 28(6), 520–5. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/15516308
Li, Q., Su, Y. A., Liu, Y., Chen, J. X., Tan, Y. L., Yang, F. De, & Si, T. M. (2014). Pharmacokinetics and tolerability of extended-release quetiapine fumarate in Han Chinese patients with schizophrenia. Clinical Pharmacokinetics, 53(5), 455–465. https://doi.org/10.1007/s40262-013-0127-9
López-Guarnido, O., Tabernero, M. J., Hernández, A. F., Rodrigo, L., & Bermejo, A. M. (2014). Rapid determination of quetiapine in blood by gas chromatography-mass spectrometry. Application to post-mortem cases. Journal of Applied Toxicology, 34(10), 1104–1108. https://doi.org/10.1002/jat.2944
Maneeton, B., Maneeton, N., Srisurapanont, M., & Chittawatanarat, K. (2013). Quetiapine versus haloperidol in the treatment of delirium: A double-blind, randomized, controlled trial. Drug Design, Development and Therapy, 7, 657–667. https://doi.org/10.2147/DDDT.S45575
Martinotti, G., Andreoli, S., di Nicola, M., di Giannantonio, M., Sarchiapone, M., & Janiri, L. (2008). Quetiapine decreases alcohol consumption, craving, and psychiatric symptoms in dually diagnosed alcoholics. Human Psychopharmacology, 23(5), 417–424. https://doi.org/10.1002/hup.944
Mattoo, S., Shah, R., Rajagopal, R., Biswas, P., & Singh, S. (2009). Quetiapine: Relatively safe in overdose? Indian Journal of Psychiatry, 51(2), 139. https://doi.org/10.4103/0019-5545.49456
Mattson, M. E., Albright, V. A., Yoon, J., & Council, C. L. (2015). Emergency department visits involving misuse and abuse of the antipsychotic quetiapine: Results from the drug abuse warning network (DAWN). Substance Abuse: Research and Treatment, 9, 39–46. https://doi.org/10.4137/SART.S22233
McConville, B., Carrero, L., Sweitzer, D., Potter, L., Chaney, R., Foster, K., … L., F. (2003). Long-term safety, tolerability, and clinical efficacy of quetiapine in adolescents: An open-label extension trial. Journal of Child and Adolescent Psychopharmacology, 13(1), 75–82. https://doi.org/10.1089/104454603321666216
McIntyre, R. S., Soczynska, J. K., Woldeyohannes, H. O., Alsuwaidan, M., & Konarski, J. Z. (2007). A preclinical and clinical rationale for quetiapine in mood syndromes. Expert Opinion on Pharmacotherapy, 8(9), 1211–1219. https://doi.org/10.1517/14656566.8.9.1211
McLarnon, M. E., Fulton, H. G., MacIsaac, C., & Barrett, S. P. (2012). Characteristics of Quetiapine Misuse Among Clients of a Community-Based Methadone Maintenance Program. Journal of Clinical Psychopharmacology, 32(5), 721–723. https://doi.org/10.1097/JCP.0b013e3182670648
Meats, P. (1997). Quetiapine (’Seroquel’); an effective and well-t oler a ted a typical antip sy cho tic. Martin Dunitz Ltd International Journal of Psychiatry in Clinical Practice, 1, 231–239.
Meisenzahl, E. M., Scheuerecker, J., Zipse, M., Ufer, S., Wiesmann, M., Frodl, T., … Möller, H. J. (2006). Effects of treatment with the atypical neuroleptic quetiapine on working memory function: A functional MRI follow-up investigation. European Archives of Psychiatry and Clinical Neuroscience, 256(8), 522–531. https://doi.org/10.1007/s00406-006-0687-x
Miodownik, C., & Lerner, V. (2006). Quetiapine: efficacy, tolerability and safety in schizophrenia. Expert Review of Neurotherapeutics, 6(7), 983–992. https://doi.org/10.1586/14737175.6.7.983
Miranda, A. S. d., Moreira, F. A., & Teixeira, A. L. (2017). The preclinical discovery and development of quetiapine for the treatment of mania and depression. Expert Opinion on Drug Discovery, 12(5), 525–535. https://doi.org/10.1080/17460441.2017.1304378
Monasterio, E., & Mckean, A. (2013). Australian and New Zealand Journal of Psychiatry. Australian & New Zealand Journal of Psychiatry, 0(0), 2012–2014. https://doi.org/10.1177/0004867412455234
Montebello, M. E., & Brett, J. (2017). Misuse and Associated Harms of Quetiapine and Other Atypical Antipsychotics. Current Topics in Behavioral Neurosciences, 34, 125–139. https://doi.org/10.1007/7854_2015_424
Morin, A. K. (2007). Possible intranasal quetiapine misuse. American Journal of Health-System Pharmacy, 64(7), 723–725. https://doi.org/10.2146/ajhp060226
Moriya, S., Tahara, Y., Sasaki, H., Hamaguchi, Y., Kuriki, D., Ishikawa, R., … Shibata, S. (2014). Effect of Quetiapine on Per1, Per2, and Bmal1 Clock Gene Expression in the Mouse Amygdala and Hippocampus. Journal of Pharmacological Sciences, 125(3), 329–332. https://doi.org/10.1254/jphs.14071SC
Müller, C., Reuter, H., & Dohmen, C. (2009). Intoxication after Extreme Oral Overdose of Quetiapine to Attempt Suicide: Pharmacological Concerns of Side Effects. Case Reports in Medicine, 2009, 1–5. https://doi.org/10.1155/2009/371698
Ngo, A., Ciranni, M., & Olson, K. R. (2008). Acute Quetiapine Overdose in Adults: A 5-Year Retrospective Case Series. Annals of Emergency Medicine, 52(5), 541–547. https://doi.org/10.1016/j.annemergmed.2008.03.016
Nishiyama, A., & Matsumoto, H. (2013). Quetiapine reduces irritability and risk of suicide in patients with agitated depression. Tokai Journal of Experimental and Clinical Medicine, 38(2), 93–96.
OBJECTIVE: To compare the pharmacology, pharmacokinetics, and efficacy of the newer atypical antipsychotics with those of conventional agents and existing atypical agents. (n.d.).
Papazisis, G., Mastrogianni, O., Chatzinikolaou, F., Vasiliadis, N., & Raikos, N. (2012). Sudden cardiac death due to quetiapine overdose. Psychiatry and Clinical Neurosciences. https://doi.org/10.1111/j.1440-1819.2012.02385.x
Parker, D. R., & McIntyre, I. M. (2005). Case studies of postmortem quetiapine: therapeutic or toxic concentrations? Journal of Analytical Toxicology, 29(August), 407–412.
Pasquini, M., Speca, A., & Biondi, M. (2009). Quetiapine for Tamoxifen-Induced Insomnia in Women With Breast Cancer. Psychosomatics, 50(2), 159–161. https://doi.org/10.1176/appi.psy.50.2.159
Pilgrim, J. L., & Drummer, O. H. (2013). The toxicology and comorbidities of fatal cases involving quetiapine. Forensic Science, Medicine, and Pathology, 9(2), 170–176. https://doi.org/10.1007/s12024-012-9404-4
Pinta, E. R., & Robert Taylor, O. E. (2007). Safety of Aripiprazole : High Serum Levels in a CYP2D6 Mutated Patient T O THE E DITOR : We present a patient with high serum levels Comment on CME Courses [ I ] wanted to pass on my thanks for this CME product . It â€TM s. I Can, 1641(January), 2007–2007. Retrieved from http://www.seeklyrics.com/
Pira, L., Mongeau, R., & Pani, L. (2004). The atypical antipsychotic quetiapine increases both noradrenaline and dopamine release in the rat prefrontal cortex. European Journal of Pharmacology, 504(1–2), 61–64. https://doi.org/10.1016/j.ejphar.2004.09.053
Pisu, C., Pira, L., & Pani, L. (2010). Quetiapine anxiolytic-like effect in the Vogel conflict test is serotonin dependent. Behav Pharmacol, 21(7), 649–653. https://doi.org/10.1097/FBP.0b013e32833e7eab
Pollak, P. T., & Zbuk, K. (2000). Quetiapine fumarate overdose: Clinical and pharmacokinetic lessons from extreme conditions. Clinical Pharmacology and Therapeutics, 68(1), 92–97. https://doi.org/10.1067/mcp.2000.107507
Potkin, S. G., Thyrum, P. T., Alva, G., Bera, R., Yeh, C., & Arvanitis, L. A. (2002). The safety and pharmacokinetics of quetiapine when coadministered with haloperidol, risperidone, or thioridazine. Journal of Clinical Psychopharmacology, 22(2), 121–130. https://doi.org/10.1097/00004714-200204000-00004
Potvin, S., Stip, E., Lipp, O., Élie, R., Mancini-Marië, A., Demers, M.-F., … Gendron, A. (2006). Quetiapine in patients with comorbid schizophrenia-spectrum and substance use disorders: an open-label trial. Current Medical Research and Opinion, 22(7), 1277–1285. https://doi.org/10.1185/030079906X112561
Quetiapine Discontinuation Syndrome. (n.d.).
Quetiapine overdose acute intoxication – case report. (n.d.).
Quetiapine overdose death – 9 case reports. (n.d.).
Rabey, J. M., Prokhorov, T., Miniovitz, A., Dobronevsky, E., & Klein, C. (2007). Effect of quetiapine in psychotic Parkinson’s disease patients: A double-blind labeled study of 3 months’ duration. Movement Disorders, 22(3), 313–318. https://doi.org/10.1002/mds.21116
Rasmussen, H., Ebdrup, B. H., Erritzoe, D., Aggernaes, B., Oranje, B., Kalbitzer, J., … Glenthoj, B. (2011). Serotonin2A receptor blockade and clinical effect in first-episode schizophrenia patients treated with quetiapine. Psychopharmacology, 213(2–3), 583–592. https://doi.org/10.1007/s00213-010-1941-5
Rasmussen, H., Ebdrup, B. H., Oranje, B., Pinborg, L. H., Knudsen, G. M., & Glenthøj, B. (2014). Neocortical serotonin2A receptor binding predicts quetiapine associated weight gain in antipsychotic-naive first-episode schizophrenia patients. The International Journal of Neuropsychopharmacology, 17(11), 1729–1736. https://doi.org/10.1017/S1461145714000777
Ray, L. A., Heydari, A., & Zorick, T. (2010). Quetiapine for the treatment of alcoholism: Scientific rationale and review of the literature. Drug and Alcohol Review. https://doi.org/10.1111/j.1465-3362.2010.00185.x
Ray, W. A., Chung, C. P., Murray, K. T., Hall, K., Michael Stein, C., & Pharmacology KTM, C. (2009). Atypical Antipsychotic Drugs and the Risk of Sudden Cardiac Death the Divisions of Rheuma- tology (. N Engl J Med, 360, 225–35.
Robert, S., Hamner, M. B., Kose, S., Ulmer, H. G., Deitsch, S. E., & Lorberbaum, J. P. (2005). Quetiapine Improves Sleep Disturbances in Combat Veterans With PTSD: Sleep Data From a Prospective, Open-Label Study. Journal of Clinical Psychopharmacology, 25(4), 387–388. https://doi.org/10.1097/01.jcp.0000169624.37819.60
Rummel-Kluge, C., Komossa, K., Schwarz, S., Hunger, H., Schmid, F., Lobos, C. A., … Leucht, S. (2010). Head-to-head comparisons of metabolic side effects of second generation antipsychotics in the treatment of schizophrenia: A systematic review and meta-analysis. Schizophrenia Research, 123(2–3), 225–233. https://doi.org/10.1016/j.schres.2010.07.012
Shaw, J. A., Lewis, J. E., Pascal, S., Sharma, R. K., Rodriguez, R. A., Guillen, R., & Pupo-Guillen, M. (2001). A Study of Quetiapine: Efficacy and Tolerability in Psychotic Adolescents. Journal of Child and Adolescent Psychopharmacology, 11(4), 415–424. https://doi.org/10.1089/104454601317261591
Shoemaker, J. M., Pitcher, L., Noh, H. R., & Swerdlow, N. R. (2003). Quetiapine produces a prolonged reversal of the sensorimotor gating-disruptive effects of basolateral amygdala lesions in rats. Behavioral Neuroscience, 117(1), 136–143. https://doi.org/10.1037/0735-7044.117.1.136
Shpaner, A., Li, W., Ankoma-Sey, V., & Botero, R. (2008). Drug-induced liver injury: hepatotoxicity of quetiapine revisited. European Journal of Gastroenterology & Hepatology, 20(20), 1106–1109. https://doi.org/10.1097/MEG.0b013e3282f8e3a0
Small, J., Hirsch, S., & Arvanitis, L. (1997). Quetiapine in patients with schizophrenia: a high-and low-dose double-blind comparison with placebo. Archives of. Retrieved from http://archpsyc.jamanetwork.com/article.aspx?articleid=497837
Smith, L. A., Cornelius, V., Warnock, A., Tacchi, M. J., & Taylor, D. (2007). Pharmacological interventions for acute bipolar mania: A systematic review of randomized placebo-controlled trials. Bipolar Disorders. https://doi.org/10.1111/j.1399-5618.2007.00468.x
Smith, R. P., Puckett, B. N., Crawford, J., & Elliott, R. L. (2004). Quetiapine overdose and severe rhabdomyolysis. J Clin Psychopharmacol, 24(3), 343. https://doi.org/10.1097/01.jcp.0000126663.04153.3f
Soeiro‑de‑Souza, M. G., Dias, V. V., Missio, G., Balanzá‑Martinez, V., Valiengo, L., Carvalho, A. F., & Moreno, R. A. (2015). Role of quetiapine beyond its clinical efficacy in bipolar disorder: From neuroprotection to the treatment of psychiatric disorders (Review). Experimental and Therapeutic Medicine. https://doi.org/10.3892/etm.2015.2213
Srisurapanont, M., Maneeton, B., Maneeton, N., Lankappa, S., & Gandhi, R. (2004). Quetiapine for schizophrenia. In Cochrane Database of Systematic Reviews. https://doi.org/10.1002/14651858.CD000967.pub2
Stein, D. J., Bandelow, B., Merideth, C., Olausson, B., Szamosi, J., & Eriksson, H. (2011). Efficacy and tolerability of extended release quetiapine fumarate (quetiapine XR) monotherapy in patients with generalised anxiety disorder: An analysis of pooled data from three 8-week placebo-controlled studies. Human Psychopharmacology, 26(8), 614–628. https://doi.org/10.1002/hup.1256
Stip, E., Fahim, C., Mancini-Mar??e, A., Bentaleb, L. A., Mensour, B., Mendrek, A., & Beauregard, M. (2005). Restoration of frontal activation during a treatment with quetiapine: An fMRI study of blunted affect in schizophrenia. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 29(1), 21–26. https://doi.org/10.1016/j.pnpbp.2004.08.015
Strickland, E. C., Cummings, O. T., Morris, A. A., Clinkscales, A., & McIntire, G. L. (2016). Quetiapine carboxylic acid and quetiapine sulfoxide prevalence in patient urine. Journal of Analytical Toxicology, 40(8), 687–693. https://doi.org/10.1093/jat/bkw078
Suttajit, S., Srisurapanont, M., Maneeton, N., & Maneeton, B. (2014). Quetiapine for acute bipolar depression: A systematic review and meta-analysis. Drug Design, Development and Therapy, 8, 827–838. https://doi.org/10.2147/DDDT.S63779
Suttajit, S., Srisurapanont, M., Xia, J., Suttajit, S., Maneeton, B., & Maneeton, N. (n.d.). Cochrane Database of Systematic Reviews Quetiapine versus typical antipsychotic medications for schizophrenia (Review) Quetiapine versus typical antipsychotic medications for schizophrenia (Review) i Quetiapine versus typical antipsychotic medications for. https://doi.org/10.1002/14651858.CD007815.pub2
Szekeres, G., Keri, S., Juhasz, A., Rimanoczy, A., Szendi, I., Czimmer, C., & Janka, Z. (2004). Role of dopamine D3 receptor (DRD3) and dopamine transporter (DAT) polymorphism in cognitive dysfunctions and therapeutic response to atypical antipsychotics in patients with schizophrenia. Am J Med Genet B Neuropsychiatr Genet, 124B(1), 1–5. https://doi.org/10.1002/ajmg.b.20045
Tarazi, F. I., Baldessarini, R. J., Kula, N. S., & Zhang, K. (2003a). Long-term effects of olanzapine, risperidone, and quetiapine on ionotropic glutamate receptor types: implications for antipsychotic drug treatment. The Journal of Pharmacology and Experimental Therapeutics, 306(3), 1145–1151. https://doi.org/10.1124/jpet.103.052597
Tarazi, F. I., Baldessarini, R. J., Kula, N. S., & Zhang, K. (2003b). Long-term effects of olanzapine, risperidone, and quetiapine on ionotropic glutamate receptor types: implications for antipsychotic drug treatment. The Journal of Pharmacology and Experimental Therapeutics, 306(3), 1145–1151. https://doi.org/10.1124/jpet.103.052597
Tarazi, F. I., Zhang, K., & Baldessarini, R. J. (2000). 【Qu-14】Olanzapine, quetiapine, and risperidone: long-term effects on monoamine transporters in rat forebrain. Neuroscience Letters, 287(2), 81–4. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/10854717
Tarazi, F. I., Zhang, K., & Baldessarini, R. J. (2002). Long-term effects of olanzapine, risperidone, and quetiapine on serotonin 1a, 2a and 2c receptors in rat forebrain regions. Psychopharmacology, 161(3), 263–270. https://doi.org/10.1007/s00213-002-1016-3
TimHill. (2006). Mundo et al. Neuropsychiatric Disease and Treatment, 2(2), 139–148.
Twaites, B. R., Wilton, L. V, & Shakir, S. A. (2007). The safety of quetiapine: results of a post-marketing surveillance study on 1728 patients in England. J Psychopharmacol, 21(4), 392–399. https://doi.org/10.1177/0269881107073257
Vaishnavi, S., Alamy, S., Zhang, W., Connor, K. M., & Davidson, J. R. T. (2007). Quetiapine as monotherapy for social anxiety disorder: A placebo-controlled study. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 31(7), 1464–1469. https://doi.org/10.1016/j.pnpbp.2007.06.027
Van den Eynde, F., Senturk, V., Naudts, K., Vogels, C., Bernagie, K., Thas, O., … Audenaert, K. (2008). Efficacy of Quetiapine for Impulsivity and Affective Symptoms in Borderline Personality Disorder. Journal of Clinical Psychopharmacology, 28(2), 147–155. https://doi.org/10.1097/JCP.0b013e318166c4bf
van der Weide, K., & van der Weide, J. (2014). The Influence of the CYP3A4*22 Polymorphism on Serum Concentration of Quetiapine in Psychiatric Patients. Journal of Clinical Psychopharmacology, 34(2), 256–260. https://doi.org/10.1097/JCP.0000000000000070
Velasco-montes, J. (2012). Clinical note Rhabdomyolysis secondary to quetiapine. Actas Esp Psiquiatr, 40(2), 97–98.
Veli, S., Atanasov, V., Angelov, J., & Kanev, K. (2014). Simultaneous application of intravenous fat emulsion and charcoal hemoperfusion in quetiapine overdose case. Open Medicine, 9(3). https://doi.org/10.2478/s11536-013-0305-z
Vieweg, W. V. R., Schneider, R. K., & Wood, M. A. (2005). Torsade de pointes in a patient with complex medical and psychiatric conditions receiving low-dose quetiapine. Acta Psychiatrica Scandinavica, 112(4), 318–322. https://doi.org/10.1111/j.1600-0447.2005.00592.x
Wang, H., Peng, Y., Tan, Q.-R., Chen, Y.-C., Zhang, R., & Qiao, Y.-T. (2010). Quetiapine Ameliorates Anxiety-Like Behavior and Cognitive Impairments in Stressed Rats : Implications for the Treatment of Posttraumatic Stress Disorder. Physiol. Res, 8408, 263–271. Retrieved from www.biomed.cas.cz/physiolres
Weisler, R. H., Montgomery, S. A., Earley, W. R., Szamosi, J., & Lazarus, A. (2012). Efficacy of extended release quetiapine fumarate monotherapy in patients with major depressive disorder. International Clinical Psychopharmacology, 27(1), 27–39. https://doi.org/10.1097/YIC.0b013e32834d6f91
Werkman, T. R., Olijslagers, J. E., Perlstein, B., Jansen, A. H. J., McCreary, A. C., Kruse, C. G., & Wadman, W. J. (2004). Quetiapine increases the firing rate of rat substantia nigra and ventral tegmental area dopamine neurons in vitro. European Journal of Pharmacology, 506(1), 47–53. https://doi.org/10.1016/j.ejphar.2004.10.053
Wiegand, M. H., Landry, F., Brückner, T., Pohl, C., Veselý, Z., & Jahn, T. (2008). Quetiapine in primary insomnia: A pilot study. Psychopharmacology. https://doi.org/10.1007/s00213-007-0968-8
Winter, H. R., Earley, W. R., Hamer-Maansson, J. E., Davis, P. C., & Smith, M. A. (2008). Steady-State Pharmacokinetic, Safety, and Tolerability Profiles of Quetiapine, Norquetiapine, and Other Quetiapine Metabolites in Pediatric and Adult Patients with Psychotic Disorders. Journal of Child and Adolescent Psychopharmacology, 18(1), 81–98. https://doi.org/10.1089/cap.2007.0084
Xu, H., Chen, Z., He, J., Haimanot, S., Li, X., Dyck, L., & Li, X. M. (2006). Synergetic effects of quetiapine and venlafaxine in preventing the chronic restraint stress-induced decrease in cell proliferation and BDNF expression in rat hippocampus. Hippocampus, 16(6), 551–559. https://doi.org/10.1002/hipo.20184
Xu, H., Wang, H., Zhuang, L., Yan, B., Yu, Y., Wei, Z., … Li, X. M. (2008). Demonstration of an anti-oxidative stress mechanism of quetiapine: Implications for the treatment of Alzheimer’s disease. FEBS Journal, 275(14), 3718–3728. https://doi.org/10.1111/j.1742-4658.2008.06519.x
Yam, M. F. C., Kiew, C. F., & Chong, C. P. (2013). Hyperglycemia and late onset seizures associated with quetiapine overdose. Tzu Chi Medical Journal, 25(2), 119–121. https://doi.org/10.1016/j.tcmj.2012.10.002
Yan, B., Bi, X., He, J., Zhang, Y., Thakur, S., Xu, H., … Li, X. M. (2007). Quetiapine attenuates spatial memory impairment and hippocampal neurodegeneration induced by bilateral common carotid artery occlusion in mice. Life Sciences, 81(5), 353–361. https://doi.org/10.1016/j.lfs.2007.05.020
Yan, B., He, J., Xu, H., Zhang, Y., Bi, X., Thakur, S., … Li, X. M. (2007). Quetiapine attenuates the depressive and anxiolytic-like behavioural changes induced by global cerebral ischemia in mice. Behavioural Brain Research, 182(1), 36–41. https://doi.org/10.1016/j.bbr.2007.05.002
Yargic, I., & Caferov, C. (2011). Quetiapine dependence and withdrawal: A case report. Substance Abuse, 32(3), 168–169. https://doi.org/10.1080/08897077.2011.558448
Young, A. C., Kleinschmidt, K. C., & Wax, P. M. (2009). Late-onset seizures associated with quetiapine poisoning. Journal of Medical Toxicology, 5(1), 24–26. https://doi.org/10.1007/BF03160977