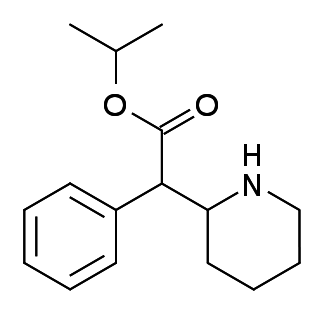
Isopropylphenidate (IPPH) is a phenidate class stimulant that appeared in the research chemical market in the 2010s. It primarily offers functional stimulation and comes with little recreational potential.
The substance is often said to be relatively good for focus and it could potentially be a good drug for those with ADHD. However, due to its research chemical status, we don’t know the drug’s long-term safety.
Isopropylphenidate = IPPH; IPH; IPPD
PubChem: 68314762
Molecular formula: C16H23NO2
Molecular weight: 261.365 g/mol
IUPAC: propan-2-yl 2-phenyl-2-piperidin-2-ylacetate
Contents
Dose
Oral
Total: 3 – 5 hours
Onset: 00:30 – 00:45
Intranasal
Total: 2 – 4 hours
Onset: 00:02 – 00:10
Timeline
Oral
Light: 5 – 15 mg
Common: 15 – 30 mg
Strong: 30 – 40 mg
Intranasal
Light: 5 – 10 mg
Common: 10 – 20 mg
Strong: 20 – 30 mg
Experience Reports
Effects
Positives
- Stimulation
- Mood elevation
- Increased focus
- Increased motivation
- Increased talkativeness
Negatives
- Increased heart rate
- Increased blood pressure
- Jaw tension
- Insomnia
- Dry mouth
- Anxiety
IPPH largely lacks recreational and euphoric potential. Most people find common doses provide little more than a minor mood boost and improved task enjoyment. Even as the dose increases, it doesn’t become an especially fun stimulant. This means it’s really geared towards productive applications, like studying and working.
Some of the intranasal use reports mention a brief euphoric “rush.”
It can be good for talking, though some people prefer to work, not talk.
The substance is clear-headed with its mental effects, allowing people to remain focused and not distracted by their own thinking or other tasks. This could stop being the case with strong+ doses, which can increase task-switching and infringe upon productivity.
Because people are more content with whatever they’re working on and they may be more motivated, it’s easier to get through boring tasks.
The focus it provides typically doesn’t lead to strong “tunnel vision,” so it’s easier to break way from a task than with some other stimulants.
It’s fairly relaxed with the way it feels physically. Users typically don’t need to move around much, sweating isn’t significant, and the cardiac activity isn’t highly noticeable. Vasoconstriction could appear, though it’s not usually uncomfortable.
Jaw tension, like with other stimulants, could appear. It seems to produce less appetite suppression than some other stimulants.
IPPH can somewhat alter your psychological status. For example, it may increase the tendency to become aggravated. And it can raise or lower anxiety, depending on the person (or have no impact).
Compared to other stimulants
The substance is often said to be more subtle than other stimulants. It can elevate your mood to a minor degree and provide some stimulation, but it doesn’t extend much beyond that.
Many people have reported it feels as good as methylphenidate from a purely functional perspective. Sometimes it may even feel better due to a relative lack of somatic effects and “speediness,” so it’s been suggested to be a particularly good ADHD drug.
Because of this effect profile, some people have already jumped to using it daily or near-daily. This isn’t recommended since it is a research chemical and little is known about its safety.
It could be lacking in the strong pro-motivation effects seen with other stimulants, though the rise in motivation may still be adequate to help people.
As long as you use a common dose, it can offer caffeine-like levels of focus enhancement and productivity, but without the somatic jitteriness.
Comedown
The comedown, if it exists, is almost always mild. There are some reports of lower motivation, lower mood, a higher likelihood of agitation, insomnia, and headache. Usually the symptoms only persist for a few hours, at most.
Chemistry & Pharmacology
Chemistry
IPPH is a member of the phenidate class of compounds, which are all esters of ritalinic acid and its analogs.
The most notable member of this class is methylphenidate. IPPH differs from that substance by the substitution of an isopropyl moiety in place of a methyl moiety.
Pharmacology
It functions as a norepinephrine-dopamine reuptake inhibitor (NDRI). This means it blocks the reuptake of dopamine and norepinephrine, effectively boosting their functional concentrations.
Compared to methylphenidate, it has significantly less impact on norepinephrine, pushing it closer to a pure dopamine reuptake inhibitor. This could potentially reduce its wakefulness effects, but it may also reduce the chance of some physical negatives like cardiovascular stimulation.
Like methylphenidate, its primary metabolite is ritalinic acid, an inactive substance. This metabolism occurs via the action of carboxylesterase 1 (CES1). IPPH seems to be a poorer substrate for CES1, which could slow down its metabolism and contribute to its lower dose range.
Papers
(Markowitz, 2013)
- Evaluated its binding using human DAT/NET/SERT in Chinese hamster ovary cells.
- Results
- Monoamine transporter binding and cellular uptake studies
- IPPH has significantly less NE activity than methylphenidate.
- Both IPPH and methylphenidate produce significant DAT inhibition, without notably affecting SERT.
- Values
- At 10 uM, the % of inhibition of control uptake
- DA uptake
- IPPH: 96%
- Methylphenidate: 90%
- NE uptake
- IPPH: 62%
- Methylphenidate: 117%
- 5-HT uptake
- IPPH: 28%
- Methylphenidate: 35%
- DA uptake
- At 10 uM, the % of inhibition of control uptake
- Hydrolysis of IPPH, ethylphenidate, and methylphenidate
- CES1 is highly expressed in human liver.
- The catalytic efficiency of CES1-mediated hydrolysis is 10-fold higher for methylphenidate than for IPPH.
- IPPH is a poor substrate for CES1. Though it’s still predominantely metabolized (hydrolyzed) by CES1, but at a markedly lower rate.
- Rat locomotor activity
- IPPH produced robust locomotor effects vs. saline following 10 mg/kg (IP).
- Monoamine transporter binding and cellular uptake studies
- Interpretation
- IPPH exhibits substantially less binding for NET than methylphenidate.
- Suggests IPPH is mainly a dopaminergic drug, with significantly less noradrenergic activity at 10 uM.
- This could lead to less impact on HR and BP.
- It’s a relatively poor substrate for CES1. Though it is primarily metabolized via this pathway to ritalinic acid, just like methylphenidate.
- Speculation: This might be due to the bulkier isopropyl substituent.
Patent US 2012/0245201
- Binding affinity at monoamine transporters
- % inhibition of control specific binding at 10 uM
- DAT
- IPPH: 97%
- Methylphenidate: 101%
- NET
- IPPH: 27%
- Methylphenidate: 88%
- SERT
- SERT: 20%
- Methylphenidate: 6%
- DAT
- % inhibition of control specific binding at 10 uM
(Luethi, 2017)
- HEK 293 cells expressing human SERT, DAT, or NET
- Results
- Monoamine uptake transporter inhibition
- IC50 for IPPH
- DAT: 0.82 uM
- NET: 2.3 uM
- SERT: 147 uM
- IC50 for methylphenidate
- IC50 for IPPH
- Monoamine uptake transporter inhibition
- DAT: 0.13 uM
- NET: 0.12 uM
- SERT: 274 uM
- Monoamine efflux
- IPPH doesn’t cause monoamine efflux.
History
1961
US patent 2012/0245201 mentions an earlier study (Portoghese and Malspeis, 1961) that suggested IPPH was more resistant to acid- and base-catalyzed hydrolysis relative to methylphenidate.
2012
US patent 2012/0245201 from John Markowitz of the Department of Pharmacotherapy and Translational Research at University of Florida, Gainesville.
This highlighted its potential utility in the treatment of ADHD and fatigue-related disorders.
It covered the use of the drug as a treatment for ADHD in children, adolescents, and adults. The dose was listed as 25 – 50 mg for dl-threo-isopropylphenidate.
March 2015
The EU’s Early Warning System was first notified about its presence. This report came from the UK.
As of March 2015
The UK’s WEDINOS drug checking organization received 7 samples of IPPH. All were white powders and the route of administration was listed as “snort/sniff.”
April 2015
Subjected to a Temporary Class Drugs Order in the UK.
As of September 2017
It’s still a member of the research chemical market.
Legal Status
US (as of September 2017)
Uncontrolled at the federal level. Always check your state laws before doing anything with the substance.
International
Australia: Uncontrolled at the federal level.
Canada: Schedule 3 as an analog of methylphenidate.
UK: Class B
Safety
The safety of isopropylphenidate isn’t known, largely due to it being a research chemical. It’s best to use common doses, infrequently, and without combinations.
An overdose could conceivably raise the risk of seizure and the risk of cardiovascular complications.
As of September 2017 there have been no fatalities linked to IPPH discussed in the literature.
Risky Combinations
- Tramadol
- Other stimulants
- Psychedelics
Test Videos
References
(2017) Pharmacological profile of methylphenidate-based designer drugs.
(2015) Temporary class drug order (TCDO) – Methylphenidate related New Psychoactive Substances (NPS)
(2015) EMCDDA–Europol 2015 Annual Report on the implementation of Council Decision 2005/387/JHA