25I-NBOMe is a psychedelic phenethylamine. It’s a potent drug with a low safety ratio, so it’s easier to encounter dangerous effects compared to LSD or psilocin.
Because it has received a lot of negative coverage in the media and has caused multiple fatalities, 25I-NBOMe doesn’t have a good reputation. Nevertheless, it’s typically safe at common doses and some people enjoy its effects.
When using strong+ doses, it may be more likely to cause confusion and be overwhelming than other psychedelics. It could also contribute to HPPD at a somewhat higher rate.
The substance was synthesized in the early 2000s, but it didn’t enter the recreational market until 2010.
Usually it’s sold on blotter paper, but it has also been sold as a powder and liquid.
25I-NBOMe = N-bomb; N-bome; Smiles; 25I; Bom-25; Cimbi-5
PubChem: 10251906
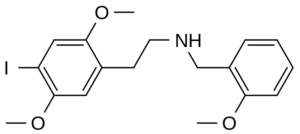
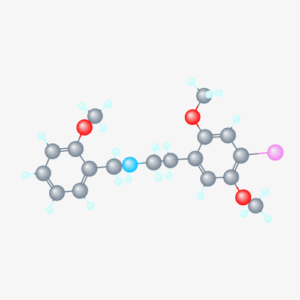
Molecular formula: C18H22INO3
Molecular weight: 427.282 g/mol
IUPAC: 2-(4-iodo-2,5-dimethoxyphenyl)-N-[(2-methoxyphenyl)methyl]ethanamine
Contents
Dose
Buccal & Sublingual
Light: 300 – 500 μg
Common: 500 – 800 μg
Strong: 800 – 1000 μg
Intranasal (tentative; not recommended)
Light: 200 – 400 μg
Common: 400 – 700 μg
Strong: 700 – 900 μg
Blotters typically range in concentration from 500 μg to 1500 μg. There can be a bit of variation in their content and you can’t necessarily trust the dosing a seller claims. As such, this is not a drug where more than one blotter should ever be taken. Typically a single blotter won’t be dangerous for an otherwise healthy person. While 2-3 blotters could conceivably be fatal, depending on the concentration.
Due to its high potency, you should avoid any intranasal use.
Timeline
Buccal & Sublingual
Total: 7 – 9 hours
Onset: 00:30 – 01:30
Intranasal (tentative; not recommended)
Total: 5 – 7 hours
Onset: 00:05 – 00:10
Experience Reports
Effects
Positive
- OEVs and CEVs
- Auditory hallucinations
- Euphoria
- Mood lift
- Stimulation – [It’s a more stimulating, energetic psychedelic. Though people can often remain seated without issue and the stimulation doesn’t negatively impact the experience.]
- Some entactogen, pro-social, or pro-empathy qualities – [These effects aren’t consistently provided, but you may notice pro-social and pro-empathy effects, especially when surrounded by other people.]
- Increased creativity
- Music enhancement
Negative
- Confusion
- Agitation
- Panic and anxiety
- Fear
- Nausea
- Thought loops
- Increased heart rate
- Increased blood pressure
- Vasoconstriction – [Leading to peripheral numbness and coldness.]
- Muscle tension
25I-NBOMe is a little shorter-acting than LSD, but it lasts longer than psilocin and 2C-B.
Perception
The visual effects are often pretty intense and can be compared to LSD overall. Dose-for-dose, it may offer more visual activity than LSD.
Significant distortions of your surroundings, trailing, fractals, geometric, and more vibrant colors are all possible.
Some people claim 25I-NBOMe’s visuals are less “natural” than those from the classic psychedelics. Not everyone reports this, however, and it’s not clear how real the difference is. It’s possible users feel that way in cases where the visual effects are prominent but the drug is lacking a strong psychedelic headspace (e.g. introspection and profoundness.)
The perceptual changes aren’t limited to the visual domain. It can also cause auditory distortions, tactile sensations, and smell changes. Synesthesia is possible.
Mental effects
Experience reports for 25I-NBOMe span the range from bliss and introspection to terror and confusion. If you stick to a common dose, it can be a fairly clear-headed psychedelic. Whereas using a strong+ dose could significantly increase the chance of confusion and even delirium.
People sometimes report strong mental effects even in the common dose range. There will be differences between individuals and even between experiences.
When confusion is present, it can present with thought loops and an inability to complete basic tasks.
Because there are reports of both strong mental activity and clear-headedness at common doses, it’s difficult to determine what the “average” 25I-NBOMe experience is like.
At strong+ doses, confusion is a much more frequent issue. You should avoid higher doses even just for that reason.
Delirium can develop when enough is used. Delirious states can be dangerous for users and those nearby. Sometimes those states are accompanied by aggression and agitation.
People have put themselves in harm’s way and even engaged in self-harm while frightened and confused because of the drug. You can reduce the chance of those negatives by using a common amount.
Occasionally people like the stronger mental activity that’s offered with high doses. It can facilitate personal insight and ego loss, for example. But if you want to explore more intense mental states, it’s best to do so with psychedelics that are physically safer.
Pleasantness
There are highly variable reports about how pleasant the experience is. This is to be expected with any psychedelic, but 25I-NBOMe might be overwhelming and dysphoric at a somewhat higher rate.
Despite that, many users report quite enjoyable and even beneficial psychedelic experiences. It can offer significant visual changes and a profound mental state, similar to LSD or psilocybin. Some users also find it can be euphoric in an LSD-like way.
It’s one of the more euphoric and mood-enhancing drugs in the NBOMe series, according to some user reports.
Negatives
Vasoconstriction is quite common. It can result in coldness and numbness, particularly in the extremities. This normally isn’t dangerous at common doses.
Muscle tension and aches are also reported fairly often.
And 25I-NBOMe can result in nausea, a generally uncomfortable tenseness and feeling of apprehension, and urinary retention (though sometimes increased urination.)
On the mental side, the greatest negatives are confusion and anxiety.
Chemistry & Pharmacology
Chemistry
25I-NBOMe is a substituted phenethylamine and a derivative of 2C-I.
Compared to 2C-I, it’s further substituted at the Nitrogen with a 2-methoxybenzyl moiety. That addition provides a higher 5-HT2A affinity and potency.
The NBOMe name for 25I and other members of that series comes from the N-Benzylmethoxy moiety and OMe (methoxy).
There are several other NBOMe derivatives where the iodine is replaced with bromine, chlorine, hydrogen, methyl, ethyl, or other substitutions. Those changes yield drugs like 25B-NBOMe, 25C-NBOMe, 25H-NBOMe, 25D-NBOMe, 25N-NBOMe, 25iP-NBOMe, and 25E-NBOMe.
N-benzyl substitutions
(Glennon, 1994) first reported on the impact of N-benzyl substitutions for phenethylamine hallucinogens with 25B-NB, a derivative of 2C-B. It was found to have a higher binding affinity than the parent drug.
It’s hypothesized the N-benzyl moiety is useful in 5-HT2A binding since the benzyl is stabilized by aromatic stacking with Phe339 in transmembrane domain 6 (TM6). Mutating Phe339 typically doesn’t impair the affinity of 5-HT2A agonists, yet it’s been found to negatively impact the activity of 25I-NBOMe and related compounds.
David Nichols’ team has found N-benzyl substitution consistently increases phenethylamine hallucinogen affinity. Though it has a greater impact if the parent compound is weaker.
For example, 2C-H receives a greater affinity boost from the substitution than 2C-I.
N-benzyl substitution paired with an oxygenated ortho position substitution can further increase 5-HT2A affinity. For example, 25I-NBOMe, 25I-NBOH, and 25I-NBMD have a higher affinity than 25H-NB.
Pharmacology
Many assays have consistently found it has a low nanomolar (nM) affinity for 5-HT2A. Functional studies suggest it has nearly full agonist properties, at least for phosphoinositide hydrolysis (the PLC-IP pathway). Most of the studies find it’s a relatively high activity partial agonist, though some have found it’s a lower-activity partial agonist.
When tested in vascular assays (e.g. rat tail artery), it was initially found to be a partial agonist. This was based on the extent of vasoconstriction, which was 30% of that from serotonin.
HTR
Animal studies have shown 25I-NBOMe produces a robust and potent head-twitch response in mice, which fits with the hallucinogenic activity seen in humans. This activity is antagonized by volinanserin, a 5-HT2A antagonist.
In this model, it was about 10-fold more potent than 2C-I, but a little less potent than LSD.
(Hanks, 2013)
- Induces the head twitch response (HTR) in mice in a dose-dependent manner.
- 0.01 mg/kg (SC) leads to a significant HTR.
- The ED50 is: 0.078 mg/kg.
- That means it’s around 14-fold more potent than 2C-I.
Functional selectivity
Some have hypothesized 25I-NBOMe could preferentially activate the PLA2-AA pathway at the 5-HT2A receptor. This could promote prostaglandin formation, thromboxane a2 formation, and inflammation.
This hasn’t been verified and the efficacy studies have generally shown high intrinsic activity based on the PLC pathway.
We also still don’t really understand how the PLA2-AA and PLC-IP properties of a given drug impact its body-wide activity. Nor do we fully understand how other signalling pathways at 5-HT2A contribute to the properties of various psychedelics.
Studies on functional selectivity have shown the parent drug, 2C-I, is somewhat biased towards IP accumulation over AA release. Other psychedelics can have quite different profiles. For example, DOI is a classic example of a high AA release psychedelic and 2C-N only seems to trigger the PLA2-AA pathway.
Clearly there’s a cause of 25I-NBOMe’s low safety ratio, but we don’t know what it is.
5-HT2A affinity and efficacy
(Ettrup, 2011)
- Antagonist binding assay
- Ki at rat 5-HT2A: 1.49 nM
- ED50 at rat 5-HT2A: 1.02 nM
- Intrinsic activity: 91%
(Ettrup, 2010)
- Found it’s a potent and selective 5-HT2A agonist.
- In vitro binding with antagonist
- Ki: 1.5 nM
- In vitro functional characterization
- Found to be an agonist in GF62 cells overexpressing 5-HT2A
- ED50: 1.02 nM at 5-HT2A
- Pretreatment with 1 uM of ketanserin fully inhibited the 25I-induced PI hydrolysis
- Intrinsic activity: 84.6%
(Braden, 2007)
- Human embryonic kidney (HEK) 293 cells expressing human 5-HT2A receptors
- Ki: 0.044 nM
- EC50: 0.44 nM
- Intrinsic activity: 81%
- Rat 5-HT2A receptor data
- Ki (antagonist): 0.087 nM
- EC50: 2.50 nM
- Intrinsic activity: 78%
(Rickli, 2015) – Actually found the N-2-methoxybenzyl substitution led to lower activation efficacy.
- Serotonin receptors (antagonist assay)
- 5-HT1A: 1800 nM
- 5-HT2A: 0.6 nM
- 5-HT2B: 130 nM
- 5-HT2C: 4.6 nM
- Serotonin receptor efficacy
- 5-HT2A EC50: 240 nM
- 5-HT2A intrinsic activity: 27% of serotonin
- 5-HT2B intrinsic activity: 32% of serotonin
- Other receptors (affinity)
- A1a adrenergic: 370 nM
- A2a adrenergic: 320 nM
- D1: 6700 nM
- D2: 900 nM
- D3: 2100 nM
- H1: 90 nM
- TAAR1 rat: 440 nM
- TAAR1 mouse: 4000 nM
- Compared to 2C-I
- Stronger binding
- A1a adrenergic
- 5-HT2a
- Weaker binding
- A2a adrenergic
- 5-HT1A
- Higher EC50 concentration for 5-HT2A and lower intrinsic activity
- Stronger binding
- Author interpretation
- N-2-methoxybenzyl substitution raises the binding affinity and/or activation potency at 5-HT2A, 5-HT2B, 5-HT2C, and adrenergic a1a receptors.
- But it lowers the binding at 5-HT1A and TAAR1.
- Receptor activation efficacy was decreased by the substitution.
- The selectivity for 2A vs. 1A was also increased, which could lead to greater effects since 5-HT1A activation seems to inhibit psychedelic activity.
- NBOMe drugs also bind to adrenergic a1 receptors, which could lead to stimulation at higher doses.
- N-2-methoxybenzyl substitution raises the binding affinity and/or activation potency at 5-HT2A, 5-HT2B, 5-HT2C, and adrenergic a1a receptors.
(Nichols, 2015)
- Human 5-HT2A affinity (antagonist)
- Ki: 0.52 nM
- Human 5-HT2A affinity (agonist)
- Ki: 0.16 nM
- Human 5-HT2C affinity (antagonist)
- Ki = 0.69 nM
- Human 5-HT2C affinity (agonist)
- Ki = 0.50 nM
- h5-HT2A
- EC50: 4.2 nM
- Emax: 86.4%
- h5-HT2B
- EC50: 15 nM
- Emax: 65%
- h5-HT2C
- EC50: 41.7 nM
- Emax: 92%
Other receptors
As you can see from the above results, 5-HT2B and 5-HT2C binding appear to exist and it operates as an agonist.
Binding data from the PDSP (antagonist assays) as reported in (Ettrup, 2010)
- 5-HT2A: 2.2 nM
- 5-HT2B: 2.3 nM
- 5-HT2C: 7.0 nM
- 5-HT6: 58 nM
- 5-HT1A: 85 nM
- D3: 117 nM
- A2C adrenergic: 348 nM
- D4: 647 nM
- Everything else is over 1 uM
The PDSP data reported in (Ettrup, 2010) shows it has the highest affinity for 5-HT2A and a similar affinity for 5-HT2B. It has a 3-fold lower affinity for 5-HT2C than 5-HT2A, based on those results. Other receptors generally show much weaker binding.
Overall, we have the most evidence for relatively selective 5-HT2A agonism. Beyond that, some of the results are conflicting. (Rickli, 2015) seems to stand apart from the other papers when it comes to the degree of intrinsic activity. Most papers support the idea that 25I-NBOMe is a high activity partial agonist, with around 80-90% activity relative to serotonin (the endogenous agonist.)
Other research
(Ettrup, 2010) – Ex vivo distribution research in rats found 25I-NBOMe uptake was greatest in the frontal cortex, moderate in striatal and thalamic regions, and low in the cerebellum.
A lipophilic metabolic was found to account for ~20% of the total plasma radioactivity, but it was only found in negligible amounts in pig brain vs. plasma.
(Catlow, 2013) – Impact on neurogenesis.
- Injections of 25I-NBOMe at 0.1, 0.3, and 1 mg/kg
- Led to a significant decline in the number of surviving BrdU+ neurons and BrdU/NeuN+ neurons in the hippocampus.
- Whereas psilocybin caused a trend towards increased neurogenesis.
(Halberstadt, 2017) – Reporting on other animal results
- 25I-NBOMe produces similar locomotor effects to those seen with other phenethylamine hallucinogens.
- 0.1 mg/kg will increase locomotor activity, while 3 mg/kg decreases it.
(Braden, 2006) – Investigation of the importance of specific amino acid residues in 5-HT2A
- In vitro tests using wild type and mutant human 5-HT2A receptor cell lines as well as NIH-3T3 cells expressing rat 5-HT2A
- Found the N-benzyl group led to significant increase in potency and affinity at r5-HT2A
- The N-benzyl analogs have their binding significantly affected with Phe339(6.51) and Phe340 (6.52) mutations within the human 5-HT2A receptor.
- Whereas classic ligands are only strongly affected by changes to Phe340(6.52).
- Phe340 mutation doesn’t appear to negatively impact intrinsic activity of 25i-NBOMe, yet it does reduce binding affinity and potency.
- Therefore, an interaction with Phe339 may be sufficient to produce a fully “active” receptor.
- These amino acid residues (Phe339 and Phe340) are within transmembrane domain 6 (TM6)
- That’s coupled to the third intracellular loop (IL3), between TM5 and TM6.
- And IL3 is important for the ability of the receptor to interact with and activate the appropriate G proteins and therefore impact second messenger production.
(Gomez-Jeria, 2015) – Molecular docking study using a model of the 5-HT2A receptor
- Important amino acid residues for 25I-NBOMe docking:
- Val156 (TMH3), Asp155 (TMH3), Ser159 (TMH3), Ile152 (TMH3), Phe339 (TMH6).
- Only two transmembrane helixes appear involved, TMH3 and TMH6.
- The only interaction common across the psychedelics tests (incl. MDMA, DMT, DOB, LSD, and mescaline) is with Val156.
Pharmacokinetics
There’s evidence that demethylation is a primary metabolic route. Some of the drugs produced could have their own activity, such as 2C-I and 25I-NBOH, though the concentrations may be too small.
CYP3A4 is thought to be the primary enzyme involved.
In vitro (human microsomal) evidence shows there is significant intrinsic clearance, which fits with extensive first pass metabolism and insufficient oral bioavailability. Most anecdotal human reports also suggest it’s not readily active when used orally.
Indeed, mice locomotor studies found SC but not IP administration could lead to activity. IP administration exposes the drug to oral-like first pass metabolism.
Given the in vitro and human toxicological data, 2C-I is one of the metabolites. Since 2C-I is much less potent, it’s probably not contributing to the drug’s activity and not enough is produced from oral administration to cause effects. This seems to be a minor metabolic route, although a fairly high 2C-I concentration has sometimes been detected.
The 2- and 5-O-desmethyl derivatives of 25I-NBOMe are rapidly conjugated with glucoronic acid, limiting their systemic exposure and potential human activity.
Papers
(Caspar, 2015)
- Tentatively identified 37 Phase 1 and 31 Phase 2 metabolites in rat urine.
- 2-O-desmethyl-25I-NBOMe, 5-O-desmethyl-25I-NBOMe, 25I-NBOH, and their glucoronic acid conjugates appear to be the primary metabolites.
- A CYP incubation screening involving ten human CYP enzymes indicated:
- CYP2C9 and CYP2C19 were involved in O-demethylation
- CYP1A2 and CYP3A4 were involved in hydroxylation
- CYP3A4 was involved in N-demethoxybenzylation
(Leth-Peterson)
- Assessed the microsomal stability of NBOMes.
- Found to have much higher clearance rates than the corresponding 2C-x drugs.
- Because 25I-NBOMe clearance (4.1 L/h/kg) exceeds hepatic blood flow (1.2 L/h/kg), extensive first pass metabolism could explain the lack of oral activity.
(Nielsen, 2016)
- In vitro metabolism study using human liver microsomes incubated with CYP enzymes and MAO enzymes.
- Metabolism in this study looking at both 25I-NBOMe and 25I-NBOH was NADPH-dependent
- Intrinsic clearance
- 4.206 L/h/kg for 25I-NBOMe
- Metabolism was 80-90% inhibited by 1-ABT, a general CYP inhibitor.
- 17 CYP-generated metabolites were identified
- Formed by hydroxylation, O-demethylation, dehydration, and N-dealkylation.
- Among the metabolites confirmed: 2C-I, 2-O-desmethyl-25I-NBOMe, 5-O-desmethyl-25I-NBOMe, 25I-NBOH, and 4-OH-25I-NBOMe.
- CYP enzymes involved
- CYP2C19 had the highest activity
- Full depletion of both drugs during incubation time.
- CYP1A2, CYP2B6, and CYP2C9 were intermediate contributors.
- While CYP3A4 could metabolize 25I-NBOMe, it didn’t metabolize 25I-NBOH.
- None of these were active: MAOA, MAOB, CYP2A6, or CYP2C18.
- And CYP2C8 and CYP2E1 only led to minor metabolite levels.
- CYP2C19 had the highest activity
- 2C-I formation
- 2C-I was found from both 25I-NBOMe and 25I-NBOH.
- CYP2C19 had the greatest ability to form 2C-I.
- CYP2B6 and CYP3A4 also formed it.
- CYP3A4
- Ketoconazole’s partial inhibition of CYP3A4 at 1 uM led to a major reduction in metabolism. 50% of starting compound survived vs. just 2%.
- When increased to a 5 uM concentration, that increased to 75% of compound remaining.
- After that inhibition, monohydroxylated and O-demethylated metabolites including 2-O-desmethyl-25I-NBOMe were still formed.
- This fits with them not being formed by CYP3A4 and it shows other CYP enzymes contribute.
- Inhibition of CYP2B6, CYP2C9, CYP2C19, and CYP2D6 also led to slightly slower metabolism, indicating smaller contribution.
- CYP2C19 appeared most important of those.
- Ketoconazole’s partial inhibition of CYP3A4 at 1 uM led to a major reduction in metabolism. 50% of starting compound survived vs. just 2%.
- Author interpretation
- Primarily metabolized by CYP3A4, but not solely by that route. There’s contribution of CYP2B6, CYP2C9, CYP2C19, and CYP2D6.
- CYP3A4 is the most abundant hepatic CYP enzyme and is also found in the gut, so metabolism in the gut could contribute to low oral bioavailability.
- When coupled with (Leth-Peterson)’s assertion that the high clearance rate exceeds hepatic blood flow, it’s understandable why there’s a lack of oral activity.
- This also suggests CYP3A4 inhibitors might make 25I more dangerous.
(Wohlfarth, 2016)
- Human hepatocytes along with in vivo mouse and human urine data
- Major Phase 1 metabolism: Demethylation, di-demethylation, and hydroxylation.
- Minor pathway is at the NBOMe ring via N-dealkylation
- Major Phase 2 metabolism: Glucuronidation, though sulfation also occurs.
- Some notable metabolites include: 2- and 5-desmethyl-25I-NBOMe and likely 5-hydroxy-25I-NBOMe
(Poklis, 2015)
- Reviewed other case reports from humans and found the parent drug is usually detected.
- 2C-I and its O-desmethyl metabolites can also sometimes be found.
- Although (Stellpflug) had a case report showing 25H-NBOMe as a urinary metabolite, that may not be a legitimate finding.
- Poklis found 25H-NBOMe was a trace contaminant in their commercial primary reference material and it’s been identified as an impurity on 25I-NBOMe blotter paper.
- Plus, dehalogenation (such as a loss of an iodine) from an aromatic ring system isn’t known to be a human drug metabolism pathway.
- 2-O-desmethyl-25I-NBOMe and 5-O-desmethyl-25I-NBOMe appear to be the best metabolites to look for in human urine samples.
- While the third demethylation metabolite, 25I-NBOH, is less abundant.
History
2003
Ralf Heim at the Free University of Berlin synthesized the drug.
His synthesis involved a reductive alkylation procedure. The starting material was 2C-I, which was reacted with 2-methoxybenzaldehyde.
This produced an imine intermediate, which could be used as a precursor. Then a reducing agent (NaBH4) was used to yield 25I-NBOMe.
2000s
Before it was used in recreational settings, 25I entered scientific study. It has primarily been explored as a tool to investigate the serotonergic system;.
Labelled 25I, namely [11C]25I-NBOMe, has been studied as a radiolabelled tracer for PET imaging.
That work continues to this day, although potentially better tracers have been identified.
Late 2000s
David Nichols’ lab at Purdue began investigating 25I-NBOMe and related compounds in the mid- to late-2000s.
They looked at their structure-activity relationships and other qualities of various NBOMe substances.
2010
Internet vendors began selling the NBOMe series and it appears 25I-NBOMe was the first to really enter the market.
It became the most popular member of the series.
2010s
25I has been sold in three primary ways:
- As a “legal” alternative to LSD. [Now outdated due to its scheduling.]
- As itself.
- Misrepresented as LSD.
There’s been a lot of incentive to sell it as LSD or in place of LSD given its lower price point. Particularly when it was legal, it could be obtained for well under $1 per dose, giving it a higher profit margin.
Some of the misrepresentation has been enabled by the use of blotters, especially in cases where the designs have historically been associated with LSD. People have often been tricked into buying and using 25I-NBOMe blotters.
A large portion of the purchases occurred online, often from dark net vendors or from Chinese vendors. It was also sold on the clearnet for a time. There’s been a lot of re-selling in the real world, often to unsuspecting buyers.
25I-NBOMe was primarily sold in blotter form, though it’s sometimes been found in powder and liquid form.
According to the EMCDDA, 25I blotters are usually pure. But a portion also contain other NBOMe drugs and/or other psychedelics.
It’s possible some of the widespread use was attributable to a large amount of media coverage. Even though the coverage was often negative (i.e. discussing fatalities), it could still increase people’s interest in the substance.
2010s
There has certainly been variation in the contents of NBOMe blotters. Buyers haven’t been able to trust the concentrations stated by sellers.
(Poklis, 2015) – Reported on an analysis of three blotters purchased online pre-late 2013
- Blotters were advertised as having 500 μg of either 25I-NBOMe, 25B-NBOMe, or 25C-NBOMe.
- Obtained through future-labs.eu.
- Results
- Every blotter’s listed drug was indeed the primary substance, but every blotter also contained some other NBOMe compounds.
- This suggests impurities are an issue.
- In the case of 25I-NBOMe, these were found at 18 – 29 ug: 25H-NBOMe, 25C-NBOMe, and 25D-NBOMe.
- Dose
- 510 μg of 25C-NBOMe on that blotter
- 540 μg of 25I-NBOMe on that blotter
- 1500 μg of 25B-NBOMe on that blotter
- Every blotter’s listed drug was indeed the primary substance, but every blotter also contained some other NBOMe compounds.
You can see from the 25B result that blotters may contain significantly more active drug than anticipated.
2010s
European seizure data was compiled by the EMCDDA at the end of 2013. The organization reported that sellers appeared to be based in the EU and China. In some cases they were offering kilograms of the substance.
The drug first showed up in the EU in May 2012. Swedish police identified the substance and reported its presence to the EU Early Warning System in June 2012.
According to a report from the EMCDDA, 14 new substituted phenethylamines were reported to the Early Warning System in 2012. 6/14 were in the NBOMe class.
Belgium
- Two cases of seizures while the drug was in transit from China to the Netherlands
- First case: 107 grams labeled as “ETHYLENE VAE”
- Second case: 51 grams labeled as “EGTAZIC ACID”
Finland
- 71 seizures during H1 2013
- 68/71 involved blotters (total number = 10,004 units)
- 3/71 involved liquid (20 ml in total)
- Total of 121 seizures from June 2012 to October 2013
- 107 at Helsinki Airport postal customs
- 14 from the police
- Typically in blotter form (n=111) with a total of 15,796 units
- Powder
- 5 cases; ranging from 0.1 to 4.5 grams
- Liquid
- 4 cases; ranging from 4 to 12 ml
- Capsules
- 1 case; with 10 capsules
Denmark
- 10 seizures of blotters (up to 263 units) between April to July 2013
- 1.5 grams of powder seized in January 2012
Germany
- 11 seizures between November 2012 and August 2013
- 2 cases of powder
- Case 1 (April 2013): 10.7 grams labeled as “1,2-Di(2-aminoethoxy)-ethaneN,N,N,Ntetraacetic acid”
- 3 cases of liquid
- All cases were in June 2012
- Range from 0.61 grams to 41.4 grams
- 6 cases of blotter
- Sometimes it was also detected in mixtures with other substances, including: 25H-NBOMe, 25C-NBOMe, and 2C-H-NBOMe.
- 2 cases of powder
Poland
- First seizure reported in 2011 in liquid form
- Mostly found as blotter; up to 391 units
- Also in powder on two occasions; 0.4 and 5.5 grams
Sweden
- May 2012 – First detection; 7 green blotters
- Blotters (5 to 583 units)
- Powders (1.49 to 13.82 grams)
Norway
- 9 seizures of blotters (1 to 250 units)
- 1 seizure of capsules (48 capsules)
France, Hungary, Italy, and the UK all reported more than five seizures.
- France
- Mainly blotters, but also 1 liquid seizure, and 2 cases of powder.
- Mostly seized by customs authorities at Roissy Airport in Paris.
- Hungary
- Small quantities of blotters (2 to 17 units)
- Powder (10.66 grams to 12.73 grams)
- Italy
- 4 to 52 blotter units
- UK
- 3 to 2,561 blotter units
Belgium, Estonia, Greece, Ireland, Portugal, and Spain each had fewer than five seizures.
2010s
Multiple deaths from the substance quickly appeared in North America, Europe, Australia, and other locations.
Between the fatalities, hospital visits, law enforcement seizures, and discussion online, the authorities and the drug-using community learned about the substance in a significant way.
October 2011
Russia became the first country to schedule the drug.
June 2011 – June 2013
The DEA reported, based on data from nationwide NFLIS labs, 795 cases of 25I-NBOMe being seized. It was typically found in powder or blotter form.
Those reports came from 35 states.
No reports existed prior to June 2011, when 25I was detected in Milwaukee, Wisconsin.
2012
Due to there being an outbreak of cases in Virginia, the state placed it in Schedule 1 .
2012
An intoxication case series in the US was presented for the first time at the Annual Meeting of the North American Congress of Clinical Toxicology.
(Kelly) reported a series involving four teenagers in Michigan who ended up in the ED due to 25I-NBOMe use.
The drug had been purchased over the internet. In 3/4 patients it caused seizures. All had tachycardia and agitation. Some of the patients required significant medical treatment.
Late 2012
(Lawn, 2014) – Non-representative internet survey of drug users (the Global Drug Survey).
- 22,289 total responses
- 2.6% (n=582) reported using an NBOMe drug
- 25I was the most popular, accounting for 2% (n=442)
- 25B was the second most popular.
- 81.2% reported using it orally or sublingually/bucally/oral
- 0.8% (n=189) reported using in the last month
- 41.7% reported sourcing from the internet.
- 39.7% reported from “a friend” and 15.9% reported from a “dealer”
- 95% reported a duration of 3-13 hours, with an average of 6 hours.
2012 – 2013
(Ezquiaga, 2016) – Examining data from samples received by Energy Control, a drug checking service in Spain.
Data was from 2009 to 2015, producing 21,198 samples in total.
- Only 56 contained 25I-NBOMe, all of which were received from 2012 onward. 2013 saw the most reports (n=19).
- Sold as:
- 42.8% as LSD
- 21.4% as 25I-NBOMe
- 7.1% as 25C-NBOMe
- 7.1% as 25I-NBOMe
- 21.4% as other substances.
February 2013 – October 2013
Austria’s drug checking program, Checkit, reported 6 cases of 25I-NBOMe detection.
5 cases involved it being sold as LSD. The other case involved it being sold as mescaline.
By 2013
32 non-fatal intoxications were reported to the EU’s Early Warning System. 15/32 were analytically confirmed.
- Belgium (3), Poland (4), Sweden (18), UK (7).
4 deaths were reported, 2 of which were analytically confirmed.
- Belgium (2), Poland (1), UK (1)
- Both analytically confirmed deaths weren’t attributed directly to 25I-NBOMe’s physical effects.
- Rather, one was “natural causes” and one was “drowning.”
- Behavioral problems could have been responsible for the latter hypothetically.
2013
The DEA reported that from March 2012 to August 2013 there were at least 19 deaths linked to 25I-, 25B-, and 25C-NBOMe.
Those people were aged 15- to 29-years-old.
June 2013
Given a temporary scheduled status in the UK.
November 2013
Emergency scheduled in the US, effective November 15, 2013.
According to the DEA, US Customs and Border Patrol had seized the drug in the form of bulk powder, often originating from Asia.
2014
As the years have gone on, there have also been instances of people receiving other NBOMe derivatives in place of 25I-NBOMe.
For example, (Laskowski, 2014) reported on two cases where people thought they took 25I-NBOMe, but they actually used 25C-NBOMe or 25B-NBOMe.
This shows an apparent evolution in the NBOMe market.
September 2014
25I-NBOMe was the most frequently reported novel psychoactive substance, according to the UN Office on Drugs and Crime.
That data came from 23 countries and territories. 43% of the regions reported the substance.
25I was followed by 25C-NBOMe and methylone. Those were found in 40% each.
2014
(Neto, 2015) – Analyzing the contents of 77 blotters from 27 street apprehensions conducted by local police forces in Minas Gerais, Brazil during 2014.
- 39 blotters from 18 cases contained an NBOMe (25I, 25B, or 25C), representing 50.6% of blotters tested and 66.7% of apprehension cases.
- Indicative of a strong presence of these drugs in the illicit psychedelic market.
2015
The DEA extended its emergency scheduling of the drug by another year.
2016
It became permanently scheduled in the US. 25I was scheduled alongside 25C-NBOMe and 25B-NBOMe.
2017
25I-NBOMe’s overall presence has seemingly declined, likely in part because of its prohibited status. However, it still shows up in the illicit market and users must remain vigilant with testing their drugs.
Legal Status
US (as of May 2017)
Schedule 1
Other countries (this list isn’t complete; always check your local laws)
Canada: Schedule 3
United Kingdom: Class A
Safety
Misrepresentation
One of the greatest issues with 25I-NBOMe is that it’s often been sold as LSD or another psychedelic. Because it’s common to take more than one blotter tab when using LSD, people could accidentally overdose on 25I with ease.
There are multiple differences between the drugs that make it relatively easy to avoid accidentally using 25I.
First, 25I blotters have a very bitter or strong chemical taste. LSD blotters can sometimes have a taste, but it’s usually nonexistent or pretty light. If you notice a very strong taste with a blotter, it may be more likely that you actually have a non-LSD drug, such as 25I-NBOMe. In cases where you notice a strong taste, you’re better off removing the blotter from your mouth and getting rid of it.
Taste is an imprecise way of determining what your blotter contains. Therefore, reagent testing is a much better option.
While also imperfect, reagent tests can at least make it significantly less likely that you’ll accidentally use 25I-NBOMe instead of LSD.
One of the best tests to use is the Ehrlich reagent. It’ll yield a purple reaction with LSD, but no reaction with 25I. You can see videos of various reagent test reactions down below.
Adverse effects
A typical overdose will include some combination of confusion, agitation, hallucinations, tachycardia, hypertension, sweating, and hyperthermia.
Those aren’t necessarily dangerous, especially if you’re treated in a medical setting.
Usually these symptoms will begin to arise in a moderate form (in other words non-fatal) when using under 2 milligrams. However, by the time you’re over 1+ mg, there’s a clear chance of severe confusion. That effect itself could be dangerous and can result in self-harm.
Severe adverse effects
More severe overdoses, typically arising from over ~2 milligrams, include a combination of: greater hypertension, agitation, thrombosis, greater tachycardia, rhabdomyolysis, seizures, metabolic acidosis, acute kidney injury, and hyperthermia.
Unlike with the average lower-level overdose, these cases require significant medical attention to avoid multi-organ failure, cardiac arrest, and other fatal outcomes.
Most of the overdose effects are largely similar to what can come from other 2C-x and DOx drugs. Although it can be easier to develop severe toxicity with 25I-NBOMe than with 2C class substances.
Fatalities
In order of how frequently they appear, these symptoms show up in severe overdoses and fatalities: seizures, rhabdomyolysis, and possibly thrombosis.
Those provide multiple routes towards organ dysfunction/failure and cardiopulmonary failure.
Serotonin syndrome
While some people have described the toxidrome as “serotonin syndrome,” that may often not be the best descriptor.
Some of the key diagnostic criteria for serotonin syndrome are: clonus and/or hyperreflexia with tremor. Those symptoms are sometimes, but not always, present in 25I-NBOMe case reports.
One of the few cases where more classic serotonin syndrome symptoms appeared involved 25I-NBOMe combined with fluoxetine, an SSRI. And there was another similar case involving a combination with sertraline, another SSRI.
Even if we don’t refer to the symptoms as “serotonin syndrome,” a serotonergic mechanism is still underlying many of the negative effects.
Problematic mechanisms
Peripheral vasoconstriction could be mediated by vascular 5-HT2A agonism. And significant peripheral vasoconstriction and vasospasm can sometimes result in limb ischemia.
The progression from rhabdomyolysis to metabolic acidosis and renal failure is a major problem in overdoses.
Disintegration of myocytes leads to the release of intracellular contents (CK, myoglobin, potassium). Those enter the plasma and can produce renal failure and cardiac failure.
Rhabdomyolysis can partly come from seizures, which are often present with severe overdoses.
Hyperthermia is believed to be connected to sympathetically mediated cutaneous vasoconstriction, which impairs heat dissipation.
Risky combinations (list may not be complete)
Other psychedelics, stimulants, tramadol, and other serotonergic drugs.
Case reviews
(Srisuma, 2015) – Data from the National Poison Data System in US
- All single-agent human exposures to NBOMe or 2C-x compounds reported to NPDS from September 2012 to September 2014 were analyzed.
- This data includes info from all of the US’ poison centers.
- 341 total cases
- 193 2C cases
- 148 NBOMe cases (138 involved 25I-NBOMe)
- Age median: 17
- Ingestion: 97
- Inhalation: 19
- Outcomes
- 49.3% had agitation/irritability
- 48.7% had tachycardia
- 40.5% had hallucinations/delusions
- 21.6% had hypertension
- 18.9% had confusion
- 15.5% had mydriasis
- 14.9% had seizures
- 9.5% had CPK elevation
- 7.4% had hyperthermia
- 7.4% had acidosis
- 6.1% had drowsiness
- 3.4% had vomiting
- 3.4% had diaphoresis
- Differences from the 2C cases
- Higher incidence of single episode seizures and hallucinations/delusions in the NBOMe group.
- Outcomes overall not deemed significantly different.
- Renal failure
- 5 cases total: 1 from NBOMe and 4 from 2C-x
- Dialysis
- 2 cases total: 1 from NBOMe and 1 from 2C-x
- Cardiac arrhythmia
- 5 cases total: 1 from NBOMe and 4 from 2C-x
- Interventions
- IV fluids, benzodiazepines, other sedatives, oxygen supplement, and intubation.
- More reports of benzodiazepine administration in the NBOMe group.
- Deaths
- 8 total: 3 from NBOMe and 5 from 2C-x
(Suzuki, 2014) – Reviewing the English literature through October 2014
- Reviewing the English literature through October 2014
- 25I-NBOMe was confirmed in 17 cases (85% of those identified to involve an NBOMe)
- 2C-I was also found in 5 cases
- Mean urine: 15.3 ng/mL (range of 2.0 to 36.0 ng/mL)
- Mean serum: 0.49 ng/mL (range of 0.034 to 0.75 ng/mL)
(Forrester, 2014) – NBOMe exposures reported to Texas Poison Centers
- Analyizing NBOMe exposures reported to Texas Poison Centers
- There were 25 total exposures; 76% involved 25I-NBOMe
- Symptoms: Tachycardia (52%), Agitation (48%), hallucinations (32%), hypertension (32%), confusion (24%), and mydriasis (20%).
- Two patients died.
- 72% only used via ingestion.
(Hill, 2013) – United Kingdom
- 7 cases in a one week period
- Case 1 involved someone getting it from a “dealer” in liquid form, which was labeled 25I-NBOMe.
- Cases 2-7 involved people at a house party who received it via the internet. Arrived via mail as a powder enclosed in purple capsules labeled “2C-B”
- Case 1
- 29-year-old male
- Presented to ED after taking 3 ml of 25I-NBOMe liquid IV
- Agitation, aggression, seizures, and self-harming behavior
- Tachycardia, hypertension, tachypnea, oxygen desaturation, and hyperthermia.
- Respiratory and metabolic acidosis, elevated creatine kinase, impaired renal function, and elevated ALT.
- Developed anuria and acute kidney injury
- Deteriorated with hyperpyrexia, high oxygen and vasopressor requirements, and worsening renal impairment.
- Day 12
- Increasingly difficult to ventilate.
- Day 17
- Improved, enabling a reduction in ventilatory support.
- Normalization of CK.
- Required very large doses of multimodal sedation during improvement period.
- Day 26
- Still anuric and CVVH dependent with creatinine peaking at 660 umol/L
- Day 27
- Able to produce urine.
- Day 38
- Left ICU
- Day 43
- Discharged
- Renal function had normalized and no apparent lasting negatives.
- Case 2
- 20-year-old male reported using 1 capsule
- Taking fluoxetine 20 mg daily as well
- Ingested the powder and then collapsed on the floor with convulsions
- Paramedic said the convulsions mainly affected his face.
- Highly agitated; required IV diazepam
- Arrived at ED
- Poor respiratory effort, clenched jaw, tachycardia, hypertension, tachypnea, urinary retention, and pupillary dilation.
- 5 hours post-admission
- Sustained clonus, ocular clonus, nystagmus, 38.6°C, tachycardia at 109, elevated creatine kinase at 228 IU/L.
- Diagnosed with serotonin syndrome.
- 12 hours post-admission
- His temperature and HR normalized
- Renal function normalized after fluid resuscitation and creatinine concentration was 95 umol/L 12 hours post-admission
- Day 2
- Respiratory status improved but jerky movements continued
- Neurologist believed they were from cerebral irritation, not seizures.
- Day 3
- Improved and extubated
- Visual hallucinations still experienced
- Day 5
- Discharged without chronic sequelae
- Case 3
- 19-year-old male reported using a “small amount” intranasally
- Almost immediately had euphoria, OEVs, CEVs, and auditory hallucinations.
- In ED
- Violent, agitated
- Required multiple doses of diazepam
- 15 hours post-ingestion
- Agitation and hallucinations resolved
- Discharged
- Case 4
- 22-year-old male
- Intranasal use
- Nausea and visual hallucinations
- Then tonic-clonic seizure which was terminated using diazepam IV.
- In ED
- Agitated and aggressive
- Required diazepam
- 15 hours post-administration
- Visual hallucinations ceased, though halos continued with eye movement.
- Creatine kinase had initially been elevated to 154 IU/L, then peaked at 633 IU/L at 12 hours, but then was 387 IU/L at 36 hours.
- Discharged on day of admission.
- Case 5
- 21-year-old male reported using 0.1 grams intranasally
- Chaotic feeling, then lost all recall until arrival at ED
- In ED
- Extremely agitated, hallucinating, violent
- Required diazepam, lorazepam, and haloperidol during the first 2 hours.
- Was tachycardic and had hyperthermia.
- 15 hours post-administration
- Symptoms resolved and was discharged.
- Case 6
- 20-year-old male reported using 1 capsule after using some alcohol
- Palpitations and then visual hallucinations
- Three inducible beats of ankle clonus, and sinus tachycardia.
- 15 hours post-administration
- Hallucinations resolved
- Discharged later that day
- Case 7
- 20-year-old male reported using 1 capsule
- Initially had a “rush” similar to ecstasy
- Then palpitations, visual and auditory hallucinations, and found it difficult to form sentences.
- Examination
- Skin was clammy, tachycardic, dilated pupils
- No nystagmus or inducible ankle clonus
- 15 hours post-administration
- Hallucinations resolved and was discharged.
- Common features
- Tachycardia, hypertension in most cases
- Hyperthermia, agitation, and seizures.
- Clonus was present in a few cases, but not all.
- Toxicology
- Plasma samples available in 7/7 cases
- 25I-NBOMe was the main active substance
- Urine available in 4/7 cases (Cases 1, 2, 6, and 7)
- 25I-NBOMe was the main active substance
- 2C-I was found in all urine samples, but none of the plasma samples.
- Interestingly, it wasn’t found in the liquid sample from Case 1, suggestive of metabolism.
- Plasma samples available in 7/7 cases
Non-fatal overdose cases
Cases reported to EU Early Warning System by 2013
- Belgium cases in August 2013 — All subjects came from the same party. — They reported using “synthetic LSD.”
- Case 1
- Lowered consciousness, insufficient breathing, mydriasis, and tachycardia.
- No other substances.
- 25I confirmed via urine.
- Case 2
- Lowered consciousness, insufficient breathing, mydriasis, and tachycardia.
- No other substances.
- 25I confirmed via urine.
- Case 3
- Headache, lessened strength in 4 extremities, mydriasis, 90 BPM, hypertension 150/85
- Symptoms disappeared after a couple hours of observation.
- No other substances.
- 25I confirmed via urine.
- Case 1
- Case 4 (UK, 2013)
- 29-year-old male
- Agitation, aggression, seizures, self-harming behavior, tachycardia 160, hypertension 187/171, tachypnea, oxygen desaturation, pyrexia, rhabdomyolysis.
- Respiratory and metabolic acidosis, elevated of creatinine kinase, impaired renal function.
- Anuria with subsequent acute kidney injury.
- Acute respiratory distress syndrome.
- Discharged from ICU on Day 38, released from hospital on Day 43.
- Other drugs: 2C-I, traces of amphetamine and methamphetamine
- 25I confirmed via urine, plasma
- Case 5 (UK, 2013)
- 20-year-old male
- Convulsions (primarily affecting face), high agitation, poor respiratory effort, clenched jaw.
- Tachycardia, hypertension, tachypnea, urinary retention, pupil dilation, pyrexia, elevated creatine kinase, visual hallucinations.
- Released from hospital on Day 4.
- Other drugs: 2C-I, traces of amphetamine and methamphetamine.
- 25I confirmed via urine, plasma
- Case 6 (UK, 2013)
- 20-year-old male
- Palpitations, visual hallucinations, pupil dilation, 3 inducible beats of ankle clonus, sinus tachycardia.
- Discharged on the day of admission.
- Other drugs: 2C-I, traces of amphetamine and methamphetamine.
- 25I confirmed via urine, plasma.
- Case 7 (UK, 2013)
- 20-year-old male
- Palpitations, visual and auditory hallucinations, tachycardia, pupil dilation.
- Discharged on the day of admission.
- Other drugs: 2C-I, traces of amphetamine and methamphetamine
- 25I confirmed via urine, plasma
- Case 8 (UK, 2013)
- 19-year-old male
- Euphoria with visual and auditory hallucinations, violent and agitated behavior
- Discharged after 15 hours.
- Other drugs: Traces of amphetamine and methamphetamine.
- 25I confirmed via plasma
- Case 9 (UK, 2013)
- 22-year-old male
- Nausea, visual hallucinations, tonic-clonic seizure, agitation, aggression, elevated creatine kinase.
- Discharged on the day of admission.
- Other drugs: Traces of amphetamine and methamphetamine.
- 25I confirmed via plasma.
- Case 10 (UK, 2013)
- 21-year-old male
- Initial chaotic feeling followed by agitation, hallucinations, violent behavior.
- Tachycardia and pyrexia.
- Discharged after 15 hours
- Other drugs: Traces of amphetamine and methamphetamine.
- 25I confirmed via plasma.
(Kelly, 2012)
- Four males between 18-19 years old
- Presented to ED after using 25I, which they got from a dealer who obtained it from the internet.
- All either intranasal or orally used it.
- Presented with tachycardia and varying levels of psychomotor agitation.
- 3/4 had prolonged seizure activity requiring drug therapy, intubation, and mechanical ventilation.
- Patient D had rhabdyomyolysis and renal failure requiring hemodialysis.
- Patient A
- HR 122
- BP 121/56
- No seizures or intubation
- 2 ng/mL in urine
- Patient B
- HR 108
- BP 140/60
- Seizures and intubation
- Patient C
- HR 153
- BP 148/49
- Seizures and intubation
- 36 ng/mL in urine
- Patient D
- HR 184
- BP 107/82
- Seizures and intubation
- 28 ng/mL in urine
(Hermanns-Clausen, 2017)
- 42-year-old male used a pediatric analgesic syrup stored in the family’s refrigerator for a severe headache
- However, the son later admitted to replacing the syrup with an ethanolic solution of 25I-NBOMe, allegedly at a 320 ug/mL concentration.
- 30 minutes post-administration
- Complained of restlessness
- Arrival at hospital
- Unremarkable findings; HR of 96 and BP of 120/80
- Dilated pupils, strong sweating, disorientation to time and to person, and agitation.
- 20 minutes post-arrival
- Severe agitation, screaming, coenesthesia, auditory and somatic hallucinations, and complex visual hallucinations (particularly serious traffic accidents)
- BP of 127/97 and HR of 100
- Lab results (incl. creatine kinase, glucose, and blood count) and clinical findings, particular temperature and ECG, weren’t remarkable.
- Therapy consisted of benzodiazepines (20 mg diazepam total) and IV fluids
- 6 hours post-arrival
- Symptoms resolved
- Discharged the next day
- Toxicology
- Sample of the “analgesic” syrup
- Unexpectedly high concentration of 2800 ug/mL
- Neither 2C-I nor 25I-NBOH were found in the liquid.
- Serum
- 50 minutes post-ingestion
- 25I-NBOMe: 34 ng/mL
- 25I-NBOH: Under 1 ng/mL
- 2C-I: 12 ng/mL
- 13 hours post-ingestion
- 25I-NBOMe: 4.2 ng/mL
- 25I-NBOH: Not detected
- 2C-I: 2.1 ng/mL
- 43.5 hours post-ingestion
- 25I-NBOMe: Not detected
- 25I-NBOH: Not detected
- 2C-I: Under 1 ng/mL
- 50 minutes post-ingestion
- Urine
- 16.5 hours post-ingestion
- 25I-NBOMe: Not detected
- 25I-NBOH: Not detected
- 2C-I: 8.2 ng/mL
- 43.5 hours post-ingestion
- 25I-NBOMe: Not detected
- 25I-NBOH: 1.2 ng/mL
- 2C-I: 3.5 ng/mL
- 16.5 hours post-ingestion
- Sample of the “analgesic” syrup
(Ameline, 2016)
- Two males discovered naked in their home
- A friend called EMS due to them acting strangely
- Both reported visual hallucinations, incoherent speech, dysarthria, and a significant aggressive behavior.
- EMS reported:
- Highly disinhibited behavior and gestures of a sexual nature (masturbation)
- Arrived at hospital 3.5 hours post-administration
- Both reported using multiple drugs ordered via internet
- Said they took methiopropamine, MDPV, amphetamines, and opiates intranasally
- Patient 1
- Coherent but highly productive speech, GSC of 15, no hallucinations, reactive bilateral mydriasis, normal cardiopulmonary examination.
- Patient 2
- Same clinical picture; primarily neurologic and psychiatric symptoms.
- Back to normal by 8.5 hours post-administration
- Discharged after 5 hours of supervision.
- Toxicology
- EMS found three small plastic bags in their home initially; two labeled 25I-NBOMe and one labeled MDPV
- Both bags with “25I-NBOMe” labels tested positive just for 25I-NBOMe.
- Two plasma samples
- 37 and 72 ng/mL of 25I-NBOMe in the men
- MDPV (59 ng/mL) was only found in Patient 1
- Patient 2 didn’t have any MDPV.
- No other drugs detected, so it was primarily a 25I-NBOMe intoxication from both.
- EMS found three small plastic bags in their home initially; two labeled 25I-NBOMe and one labeled MDPV
(Malakooti, 2013)
- 16-year-old male
- Found seizing
- Presented and successfully treated seizure activity with benzodiazepines
- Persistence of altered mental status; combativeness and agitation.
- Lab: Thrombocytopenia, coagulopathy, acute renal failure, acute lung injury, lactic acidosis, and elevated creatine kinase due to severe rigors.
- Intubated
- Dexmedetomidine given to address serotonin syndrome symptoms via central alpha blockade
- Good results from this.
- Over the next 24 hours
- Developed hemodynamic lability with impaired oxygen utilization, progressive lactic acidosis, and a narrow AVDO2. Plus worsening oxygen dysfunction.
- Given thiamine, L-carnitine, and CoQ10 to address metabolic uncoupling and to promote oxygen utilization.
- Lactate decreased and AVDO2 began to reach physiologic levels.
- Plasmapheresis performed to deal with coagulopathy and to remove residual 25I bound to plasma.
(Hieger, 2012)
- Ten patients with avg age of 17 (range=14 to 20)
- Presented to local EDs following ingestion and/or intranasal use of “25-I”
- 6/10 only reported 25I use, while the others reported ethanol, 2C-E, THC, and/or ketamine.
- Most common effects
- Tachycardia (90%), Hypertension (70%), agitation (60%), and hallucinations (50%)
- Average heart rate: 123 (range of 78 – 150s)
- 2 patients found in status epilepticus and another 1 patient found unresponsive
- 1/2 patients with a seizure also had multiple, discrete intraparenchymal hemorrhages and acute kidney injury.
- Hyperthermia wasn’t present in any of the cases.
- 60% admitted to ICU, 20% treated in ED and released, 1 admitted to psychiatry, 1 managed in clinical decision unit.
- 3/10 required emergent intubation and all got IV benzodiazepines for sedation.
- All were discharged once they were in good condition.
- One agitated and tachycardic patient had their blood analyzed
- Revealing serum 25I-NBOMe concentration: 0.76 ng/mL
(Rose, 2013)
- 18-year-old male presented to ED with severe agitation and hallucinations after jumping out of a moving car.
- Tachyardiac 150, BP 150 to 170/110
- Required physical restrains and treatment with IV lorazepam
- Gradually improved and vital signs returned to normal over 48 hours, though he continued to have episodes of aggression.
- Serum concentration (during ED evaluation and treatment):
- 0.76 ng/mL
(Stellpflug, 2013)
- 18-year-old female
- Reported to ED after tonic-clonic seizure following self-reported use of 25I sublingually
- She only reported using one dose.
- Seizure occurred ~90 min after administration. Seizure activity stopped by the time the paramedics arrived.
- Presented with tachycardia, hypertension, confusion, and agitation.
- HR of 145, BP of 145/100
- Cutaneous flushing and hyperreflexia
- Improved with IV fluids and benzodiazepines
- Urine:
- 25I-NBOMe = 7.5 ng/mL
- Desmethyl-25I-NBOMe = presumptive at 600 ng/mL (meaning it was 80-fold higher than the unchanged drug in urine)
- 25H-NBOMe = 0.9 ng/mL (extrapolated below the LLOQ)
- 2C-I = 1.8 ng/mL
- 25H-NBOMe hypothesized to be an impurity from the synthesis since the authors couldn’t think of a way it’d arise from metabolism.
(Poklis, 2014)
- Three patients had presence of 25I-NBOMe after presenting to ED
- Symptoms: tachycardia, hypertension, severe agitation, seizures.
- One case only detected 25I at 0.1 ng/mL
- Another case had 25I (2.3 ng/mL) and 25C-NBOMe
- Final case had 25I-NBOMe (1.2 ng/mL) and 25H-NBOMe
(Suzuki, 2014) – Self-harming behavior
- 18-year-old male
- Brought to ED after calling 911 reporting he had tried to commit suicide after taking “two hits of acid.”
- College freshman w/ no history of suicidal ideation or psychiatric diagnosis. Though he had 3-4 months of mild depression in the context of stressors at school, yet he denied any suicidal ideation.
- Interest in trying LSD and got “good LSD” from his friend.
- Upon taking the two blotters, he noted a moderately bitter taste.
- Effects built throughout the night, leading to greater confusion and anxiousness.
- He retreated to his dorm room and felt “trapped.”
- As his anxiety worsened, he began to experience panic and started contemplating suicide to end the experience.
- Proceeded to use a paid or scissors to stab his neck and chest.
- Unable to remember the events following the self-harm.
- ~11 hours after using “LSD”
- Realized extent of injuries and called 911.
- Arrived to ED alert, anxious, and in moderate distress. Reported no longer being under the drug’s influence.
- Afebrile, HR of 90, BP of 140/84, and respiration of 20.
- Notable injuries were found in the indicated regions.
- Serum sample (from admission): 25I-NBOMe at 34 pcg/mL
(Richeval, 2017) – Case report and examination of metabolites
- 29-year-old male
- Also using sertraline
- And history of generalized anxiety disorder and transient LSD-induced psychotic symptoms
- Experienced acute unconsciousness within 1 hour of intranasally using a drop of pink liquid
- Examination
- Hypertonia, tremors, partial seizure followed by generalization, mydriasis, tachycardia 120, hypertension 225/70, hyperthermia 39°C, and profuse sweating.
- Symptoms indicative of serotonin syndrome
- Improved with diazepam and clonazepam
- Transferred to ICU
- Presented with neurological worsening and quiet coma interspersed with tonic movements requiring sedation and aspiration pneumonitis.
- Invasive ventilation for 24 hours
- High creatinine, high AST, high ALT.
- Discharged on Day 11 with persisting confusion and memory disorder.
- Day 240 follow-up
- Persistent cognitive and psychiatric abnormalities, with temporospatial disorientation, memory impairment, and dysexecutive syndrome, requiring antipsychotic treatment.
- Toxicology
- Pink liquid sample
- Confirmed for 25I-NBOMe as the main drug, along with 25H-NBOMe and 25C-NBOMe.
- Serum
- 3 hours post-admission
- 25I-NBOMe: 0.9 ug/L
- No metabolites
- 15 hours post-admission
- 25I-NBOMe: Not detected
- No metabolites
- 3 hours post-admission
- Urine
- 3 hours post-admission
- 25I-NBOMe: 1.4 ug/L
- Desmethyl-25I-NBOMe (1): 3.36 ug/L
- Desmethyl-25I-NBOMe (2): 0.54 ug/L
- Desmethyl-hydroxy-25I-NBOMe: 0.30 ug/L
- Hydroxy-25I-NBOMe: 0.34 ug/L
- Di-desmethyl-25I-NBOMe: 0.81 ug/L
- Desmethyl-25I-NBOMe glucuronide: 2.90 ug/L
- Hydroxy-25I-NBOMe glucuronide: 0.16 ug/L
- 15 hours post-admission
- 25I-NBOMe: 1.2 ug/L
- Desmethyl-25I-NBOMe (1): 3.50 ug/L
- Desmethyl-25I-NBOMe (2): 0.35 ug/L
- Desmethyl-hydroxy-25I-NBOMe: 0.19 ug/L
- Hydroxy-25I-NBOMe: 0.21 ug/L
- Di-desmethyl-25I-NBOMe: 0.43 ug/L
- Desmethyl-25I-NBOMe glucuronide: 3.08 ug/L
- Hydroxy-25I-NBOMe glucuronide: 0.50 ug/L
- 3 hours post-admission
- Pink liquid sample
Fatalities
Cases reported to EU Early Warning System by 2013
- Case 1 (Belgium, 2013)
- Male
- “Natural” cause of death.
- No other drugs reported.
- 25I confirmed.
- Case 2 (UK, 2013) reported in (Soh, 2014)
- 22-year-old male
- Drowning was the cause of death.
- Other drugs: Amphetamine, ketamine, lidocaine, 5-MeO-DiPT, DOI, 25C-NBOMe, 2C-I.
- 25I confirmed via post-mortem blood.
DEA obtained medical examiner and postmortem toxicology reports from various states implicating some combo of 25I, 25C, and 25B-NBOMe in the death of 14 people.
- Average age: 20 (range of 15 to 29)
- 11: Acute toxicity as cause of death
- 3: Violent behavior as cause of death
- June 2012 (within series)
- Two teenagers fatally overdoses on a substance that was allegedly 25I-NBOMe and a
- October 2012 (within series)
- A 21-year-old man died of an apparent overdose after taking a liquid drop of 25I-NBOMe nasally at a music festival.
- January 2013 (within series)
- 18-year-old died after taking 25I-NBOMe sold as LSD.
- Toxicology screen ruled the cause of death as 25I-NBOMe acute poisoning.
(Walterscheid, 2014) – United States
- 2 deaths stemming from 25I-NBOMe use at rave parties.
- Case 1
- 21-year-old male drive who admitted to taking “acid” to the passenger.
- Two “hits of acid” were taken after the rave party. Cannabis was also used.
- He suddenly had a surge of violent behavior.
- He began to hallucinate and flail about, striking his friend and ripping the stretched piercing jewelry out of his own earlobes.
- Friend helped pull the vehicle to the side of the road while the decedent continued to punch the console and rip accessories off of the interior of the vehicle.
- The friend exited the vehicle; searched for road sign while calling emergency services
- Return to find decedent unresponsive in the front of the vehicle.
- Friend tried to perform cardiopulmonary resuscitation; yet decedent was pronounced dead upon arrival of emergency services.
- Scene examination showed vomit in the car and damage to the vehicle.
- Tox screens confirmed 25I-NBOMe use (heart blood and urine samples)
- Also cannabis metabolite.
- 21-year-old male drive who admitted to taking “acid” to the passenger.
- Case 2
- 15-year-old female
- Socializing outside a rave party and eventually used an unknown clear liquid orally.
- While being driven home by a friend, the decedent complained about not feeling well, began flailing her arms and legs, and then became unresponsive.
- Driven to local hospital
- Presented with asystole and temp of 39.9°C.
- Intubated and advanced cardiac life support began.
- Pronounced dead ~18 minutes after arrival.
- Tox screens confirmed 25I-NBOMe use (heart blood and urine)
- Also cannabis metabolite.
(Oosthuizen, 2016) – Providing information during the 2016 TIAFT meeting
- 23-year-old female
- Intranasally used an unknown white powder believed to be “synthetic LSD”
- Toxicology
- Aortic blood
- 28 ng/mL of 25I-NBOMe
- 1 ng/mL of 25H-NBOMe
- 0.7 ng/mL of 25C-NBOMe
- Aortic blood
(Kueppers, 2015) – Australia
- 23-year-old female
- Seen intranasally using an unknown substance believed to by “synthetic LSD”
- Witnesses said she used a line approximately 10 mm long and 1 mm wide
- Shortly after:
- Began acting strangely, uttering random words, appearing agitated, and clinging onto her boyfriend’s arm.
- She kept intermittently shouting random words.
- Eventually she started thrashing about aggressively, before apparently seizing and vomiting, following by collapse and death.
- Cardiopulmonary resuscitation attempts were unsuccessful.
- Postmortem exam
- Confirmed congestion of the lungs and the lungs showed mild pulmonary edema.
- Toxicology
- Confirmed 25I-NBOMe, 25H-NBOMe, and 25C-NBOMe
- Also confirmed methamphetamine, THC, and a small amount of promethazine.
- No alcohol or other drugs.
- Aortic blood
- 25I-NBOMe was easily the highest concentration: 28 ug/L
- 25H-NBOMe: 1 ug/L
- 25C-NBOMe: 0.7 ug/L
- The methamphetamine was at 0.39 mg/L, considered to be in the lower range of reported normal “recreational” levels and not likely to be fatal on its own.
- 25I considered the primary cause of death.
(Poklis, 2013)
- 19-year-old male
- He was initially trip-sitting for friends and then ingested blotter paper containing “acid.”
- First time he had ever used the drug or any psychedelic.
- Whereas the other users didn’t report his effects but had experience.
- Friends noticed his strange and paranoid behavior; agreed it’d be wise to change location and go for a walk.
- His behavior become more bizarre and he abruptly walked away from his friends.
- Friends eventually went to his apartment searching for him; found him prone and unresponsive on the pavement near the apartment complex’s swimming pool.
- He had either jumped or fallen off his apartment balcony located multiple stories up.
- 911 called; he was pronounced dead at the scene
- Toxicology
- Peripheral whole blood: 405 pg/mL of 25I-NBOMe which is within the range from other intoxicated patients.
- Aftermath
- Supplier of the “acid” was arrested after the death.
- He told police 25I-NBOMe was used; had been purchased online in powder form.
- Claimed 1 gram would make 2000 “hits”
- If that’s the concentration that was employed, that’d yield 500 μg per dose.
- Gastric contents from decedent only showed one piece of white blotter paper, indicating he had taken around 500 μg.
(Shanks, 2015)
- 16-year-old male
- Partying with friends and used a drug on a blotter paper.
- Found unresponsive by friends in the morning.
- EMS called; he was pronounced dead
- Autopsy
- Severe and diffuse pulmonary edema
- Mild cerebral edema
- Toxicology
- Heart blood: 25I-NBOMe at 19.8 ng/mL
- Urine: Positive for 25I-NBOMe
- No other drugs found in the blood or urine.
(Lowe, 2015) – Combination of 25I-NBOMe with other drugs, though it was likely a primary contributor.
- 15-year-old male
- Went to a party in August 2014 and the DJ was reportedly selling mushrooms and “25I (acid)”
- Decedent seen taking mushrooms and drinking a vial of unknown clear liquid
- Hour later
- Vomiting and convulsion
- Still coherent enough to say, “This is awesome.”
- Laughing and telling people was fine
- But then he continued to have more convulsions until he lost consciousness.
- Transported to fire station; failed resuscitation efforts.
- Transferred to local hospital
- Intubated
- Acute renal failure with serum creatine of 4 mg/dL
- Liver failure with AST and ALT levels in the 10,000s
- Coagulopathic w/ disseminated intravascular coagulation-like presentation.
- Died from multi-system organ failure following cardiopulmonary arrest 3 days post-administration.
- Toxicology
- Blood
- Positive for 25I-NBOMe at 0.76 ng/mL
- Urine
- Positive for 25I-NBOMe, 25C-NBOMe, and 25H-NBOMe.
- Also positive for psilocin.
- Blood
(2014)
- 17-year-old female
- She was on lithium (potentially serotonin syndrome contributor) and topiramate for mood disorders
- Ingested an unknown substance on blotter paper.
- Presented
- Status epilepticus, hyperthermia, hypokalemia, hypocalcemia, elevated transaminases, acute kidney injury, metabolic acidosis, and rhabdomyolysis.
- Subsequently developed cerebral edema and was then declared brain dead on Day 7 in the hospital.
- Toxicology
- Antemortem
- Whole blood: 25I-NBOMe at more than 0.25 ng/mL
- Lithium at 0.34 mmol/L (lower than the therapeutic range)
- Antemortem
- Interpretation
- Even though the lithium was at a low concentration, this case was considered to be a death from serotonin syndrome resulting from 25I-NBOMe and lithum.
Test Results
References
(2017) Bad trip due to 25I-NBOMe: a case report from the EU project SPICE II plus.
(2017) Identification of 25I-NBOMe in two intoxications cases with severe hallucinations
(2017) Pharmacology and Toxicology of N-Benzylphenethylamine (“NBOMe”) Hallucinogens.
(2017) Retrospective identification of 25I-NBOMe metabolites in an intoxication case
(2016) 25I-NBOMe: The legal LSD
(2015) A Note on the Docking of some Hallucinogens to the 5-HT2A Receptor.
(2015) Fatal Intoxications with 25B-NBOMe and 25I-NBOMe in Indiana During 2014.
(2015) NBOMe: a new dangerous drug similar to LSD
(2015) A Case Review of the First Analytically Confirmed 25I-NBOMe-Related Death in Washington State.
(2015) Evolution of the NBOMes: 25C- and 25B- Sold as 25I-NBOMe.
(2015) 2C-I-NBOMe, an “N-bomb” that kills with “Smiles”. Toxicological and legislative aspects.
(2015) NBOMe: new potent hallucinogens–pharmacology, analytical methods, toxicities, fatalities: a review.
(2015) Prevalence of use and acute toxicity associated with the use of NBOMe drugs.
(2015) 25I-NBOMe related death in Australia: A case report
(2015) NBOMe and 2C substitute phenylethylamine exposures reported to the National Poison Data System.
(2015) Rapid detection of NBOME’s and other NPS on blotter papers by direct ATR-FTIR spectrometry.
(2014) EMCDDA–Europol Joint Report on a new psychoactive substance: 25I-NBOMe
(2014) NBOMe designer drug exposures reported to Texas poison centers.
(2014) Lithium- Fatal toxicities when combined with 25I-NBOMe: case report
(2014) New psychoactive substances as adulterants of controlled drugs. A worrying phenomenon?
(2014) Pathological findings in 2 cases of fatal 25I-NBOMe toxicity.
(2014) 25I‐NBOMe Critical Review Report
(2013) The NBOMe series: a novel, dangerous group of hallucinogenic drugs.
(2013) A case of 25I-NBOMe (25-I) intoxication: a new potent 5-HT2A agonist designer drug.
(2013) NBOMe — a very different kettle of fish
(2013) Multi-organ failure associated with ingestion of synthetic street drug 25I
(2010) Radiosynthesis and evaluation of 11C-CIMBI-5 as a 5-HT2A receptor agonist radioligand for PET.